Welcome! We are the Bovine Satellite Cell (BSC) Immortalization group. We’re working in the Kaplan Lab at Tufts University to genetically modify cow muscle cells such that they cannot senesce and die of old age, as most cells do. Our goal is to produce cells that can one day be used to study and produce cultured meat, to provide an ecologically and ethically sustainable alternative to factory farming.
Why Cultured Meat?
The biggest motivator behind this project is climate change. Food production currently accounts for approximately 26% of all greenhouse gas emissions, and meat, one of the most desired types of food worldwide, is unfortunately by far the most polluting food group in existence. This is especially true of beef, which produces almost twice as much greenhouse gas per kilogram of food as its next-closest competitor, lamb. As the world becomes more affluent and demand for meat continues to rise, this is only going to become a bigger problem for the environment, so something needs to be done to alleviate this burden or the consequences for our planet will be dire.
This project is also being undertaken for ethical reasons. Vegetarianism and veganism are on the rise throughout the developed world, and one of the most commonly cited reasons that people make the switch is learning about the awful living conditions of animals in factory farms. These farms aren’t going anywhere in the conventional meat production industry; they’re simply the most cost-effective way of keeping up with global demand while continuing to use traditional livestock. Our team aims to get around this problem, and give people a way to eat real meat-based products without feeling like they have to compromise their morals to do so.
Why Immortalize BSCs?
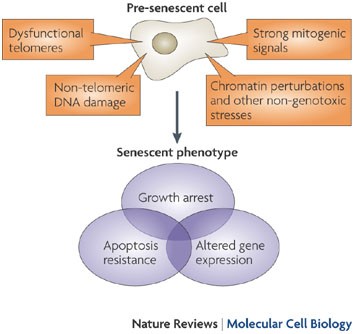
At the moment, arguably the greatest problems facing the field of cultured meat are cost and scale-up potential. Simply put, there currently isn’t a widely known way of growing muscle cells quickly or cheaply enough that they could provide a realistic alternative to conventional meat sources, and one of the most significant reasons for this is cellular senescence. This is what happens when cells get too old and become damaged: they stop dividing, change many of their biological properties, and eventually die. This happens all the time in cultured meat production, and it means that not only do researchers need a constant source of fresh bovine stem cells (which generally involves sacrificing young cows, mitigating the ethical benefits of cultured meat), but also that growing cells to any sort of large scale tends to be very difficult and very expensive.
Our team aims to address this problem by producing bovine cells that can continue to grow and divide forever. This would deal with both the sourcing and price challenges above, which would be highly significant to future research and development projects in the cultured meat field.
How Are We Going to Do This?

You can read a lot more about our process in the Project Overview section of this webpage, but the general theme of the project is trial and error with genetic engineering. We have identified some key genes that are able to prevent senescence in other cell types, so we’re going to be attempting to integrate them into our BSCs using several different gene editing methods. Some of these are quite widely known, like CRISPR-Cas9, others less so, like the Sleeping Beauty transposon system (amusingly named after a fish, not the Disney princess). You can take a look at the overall flow of our project above.
In summary, cultured meat has the potential to make a massive impact on the environmental and ethical side effects of meat consumption, but it currently struggles with cost and scalability. Our mission is to address these challenges by using genetic engineering to produce cow muscle cells that can divide forever, giving researchers and startups a potent new tool to accelerate their projects. For further details and clarification, please examine the rest of this website at your leisure, and feel free to contact us at the emails listed on this page.