Brian Timko’s research interests lie at the intersection of materials science, chemistry and biology, with a major focus on nanotechnology and nanoscale interfaces between solid-state and biological systems. Central to our laboratory’s strategy is the bottom-up paradigm, whereby nanomaterials are synthesized with rationally-controlled electrical, optical and chemical properties, then assembled into biological systems at physiological conditions. This strategy is leveraged to achieve advances in three interrelated areas:
Nanoelectronic interfaces with cells and tissues
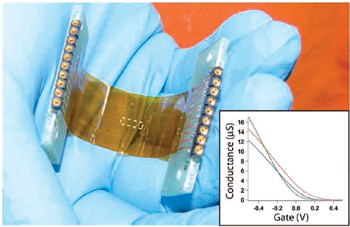 Semiconductor nanowires can be elucidated as field-effect transistors (NW-FETs), which function as highly-sensitive detectors of local electric field changes. NWs are synthesized via the vapor-liquid-solid growth technique, which enables rational control over geometry and chemical composition. These nanomaterials are assembled onto biocompatible substrates, then individually addressed with lithographically-defined metallic interconnects. Functionalized with specific antibodies or other receptors, NW-FETs achieve specific, rapid biological detection. We are now exploring methods by which to stably interface NW-FETs with living cells and tissues, to achieve extra- or intracellular electrophysiology, or to monitor chemical signals in real time.
Semiconductor nanowires can be elucidated as field-effect transistors (NW-FETs), which function as highly-sensitive detectors of local electric field changes. NWs are synthesized via the vapor-liquid-solid growth technique, which enables rational control over geometry and chemical composition. These nanomaterials are assembled onto biocompatible substrates, then individually addressed with lithographically-defined metallic interconnects. Functionalized with specific antibodies or other receptors, NW-FETs achieve specific, rapid biological detection. We are now exploring methods by which to stably interface NW-FETs with living cells and tissues, to achieve extra- or intracellular electrophysiology, or to monitor chemical signals in real time.
– Read our review about nanoelectronic interfaces with biological systems.
Nanocomposite materials for tissue engineering
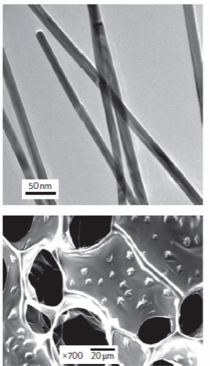 The extracellular matrix (ECM) plays a crucial role in tissue engineering, providing requisite structural and biochemical cues for tissue morphogenesis and homeostasis. Nanomaterials embedded within tissue scaffolds can recapitulate the ECM by modulating electrical and mechanical properties and also by providing binding sites, e.g. for focal adhesion formation. For example, lyophilized alginate constructs containing gold nanowires were effective scaffolds for cardiac tissue engineering. Compared to their pristine counterparts, nanocomposite scaffolds supported tissues with higher expression of connexin-43 and actinin filaments, and substantially improved electrical and mechanical synchrony. We are actively developing 3D bioprinting techniques to achieve new classes of nanocomposite tissues with tunable properties and embedded vasculature.
The extracellular matrix (ECM) plays a crucial role in tissue engineering, providing requisite structural and biochemical cues for tissue morphogenesis and homeostasis. Nanomaterials embedded within tissue scaffolds can recapitulate the ECM by modulating electrical and mechanical properties and also by providing binding sites, e.g. for focal adhesion formation. For example, lyophilized alginate constructs containing gold nanowires were effective scaffolds for cardiac tissue engineering. Compared to their pristine counterparts, nanocomposite scaffolds supported tissues with higher expression of connexin-43 and actinin filaments, and substantially improved electrical and mechanical synchrony. We are actively developing 3D bioprinting techniques to achieve new classes of nanocomposite tissues with tunable properties and embedded vasculature.
– Read our review about nanotechnological strategies in tissue engineering.
Embeddable Electronics for Hybrid Tissues
Hybrid tissues – that is, constructs consisting of engineered tissues with embedded electronic components – could enable fundamentally new studies of tissue function with applications in neuroscience, pharmacology or regenerative medicine. We are developing soft, conformal materials that will enable us to embed devices and interconnects within innervated tissue models in 3D configurations. Concurrently, we are exploring methods to (a) achieve high yields of neuron/device interfaces, (b) record and process data in a highly-multiplexed fashion and (c) apply data analysis techniques to abstract useful information about the state of the tissue.
Nanoelectronic interfaces with cells and tissues
 Semiconductor nanowires can be elucidated as field-effect transistors (NW-FETs), which function as highly-sensitive detectors of local electric field changes. NWs are synthesized via the vapor-liquid-solid growth technique, which enables rational control over geometry and chemical composition. These nanomaterials are assembled onto biocompatible substrates, then individually addressed with lithographically-defined metallic interconnects. Functionalized with specific antibodies or other receptors, NW-FETs achieve specific, rapid biological detection. We are now exploring methods by which to stably interface NW-FETs with living cells and tissues, to achieve extra- or intracellular electrophysiology, or to monitor chemical signals in real time.
Semiconductor nanowires can be elucidated as field-effect transistors (NW-FETs), which function as highly-sensitive detectors of local electric field changes. NWs are synthesized via the vapor-liquid-solid growth technique, which enables rational control over geometry and chemical composition. These nanomaterials are assembled onto biocompatible substrates, then individually addressed with lithographically-defined metallic interconnects. Functionalized with specific antibodies or other receptors, NW-FETs achieve specific, rapid biological detection. We are now exploring methods by which to stably interface NW-FETs with living cells and tissues, to achieve extra- or intracellular electrophysiology, or to monitor chemical signals in real time.
– Read our review about nanoelectronic interfaces with biological systems.
Nanocomposite materials for tissue engineering
 The extracellular matrix (ECM) plays a crucial role in tissue engineering, providing requisite structural and biochemical cues for tissue morphogenesis and homeostasis. Nanomaterials embedded within tissue scaffolds can recapitulate the ECM by modulating electrical and mechanical properties and also by providing binding sites, e.g. for focal adhesion formation. For example, lyophilized alginate constructs containing gold nanowires were effective scaffolds for cardiac tissue engineering. Compared to their pristine counterparts, nanocomposite scaffolds supported tissues with higher expression of connexin-43 and actinin filaments, and substantially improved electrical and mechanical synchrony. We are actively developing 3D bioprinting techniques to achieve new classes of nanocomposite tissues with tunable properties and embedded vasculature.
The extracellular matrix (ECM) plays a crucial role in tissue engineering, providing requisite structural and biochemical cues for tissue morphogenesis and homeostasis. Nanomaterials embedded within tissue scaffolds can recapitulate the ECM by modulating electrical and mechanical properties and also by providing binding sites, e.g. for focal adhesion formation. For example, lyophilized alginate constructs containing gold nanowires were effective scaffolds for cardiac tissue engineering. Compared to their pristine counterparts, nanocomposite scaffolds supported tissues with higher expression of connexin-43 and actinin filaments, and substantially improved electrical and mechanical synchrony. We are actively developing 3D bioprinting techniques to achieve new classes of nanocomposite tissues with tunable properties and embedded vasculature.
– Read our review about nanotechnological strategies in tissue engineering.
Embeddable Electronics for Hybrid Tissues
Hybrid tissues – that is, constructs consisting of engineered tissues with embedded electronic components – could enable fundamentally new studies of tissue function with applications in neuroscience, pharmacology or regenerative medicine. We are developing soft, conformal materials that will enable us to embed devices and interconnects within innervated tissue models in 3D configurations. Concurrently, we are exploring methods to (a) achieve high yields of neuron/device interfaces, (b) record and process data in a highly-multiplexed fashion and (c) apply data analysis techniques to abstract useful information about the state of the tissue.