Management of Heart Failure
John E. Rush, DVM, MS, DACVIM (Cardiology), DACVECC
Cummings School of Veterinary Medicine At Tufts University
Last Updated 03/30/2020
OBJECTIVES
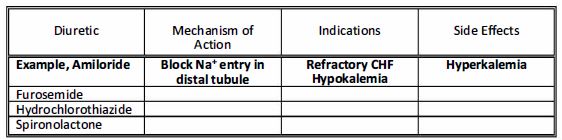
Diuretics
1. Select your first choice diuretic drug for treatment of heart failure.
2. Select a second diuretic to add to your first for management of refractory CHF.
3. List the mechanism of action, the indications and possible side effects for the following diuretics.
Positive lnotropes
1. List 2 ways that digoxin might be useful in treatment of heart failure.
2. List the arrhythmias that might be improved following digoxin therapy.
3. A dog with renal failure and clinical signs of digoxin toxicity is presented for your evaluation. What clinical signs lead you to suspect digoxin toxicity? How would you confirm your suspicion of digoxin intoxication? Predisposing factors?
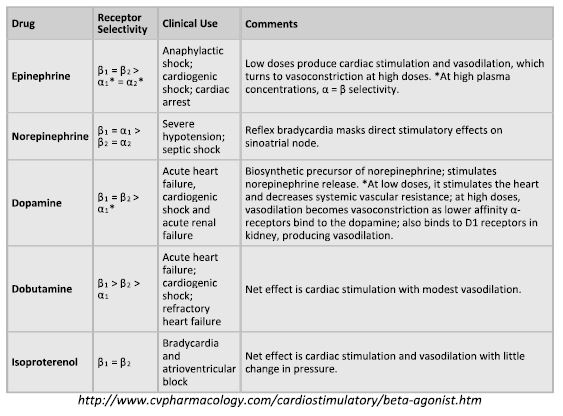
4. Outline the a, B-1 and B-2 effects of:
•Epinephrine •Norepinephrine •Dopamine •Dobutamine • lsoproterenol
5. Your technician calls you over to assist with a cat that has just developed cardiopulmonary arrest with asystole. What drug will you give? What dosage? What route? What effect(s) are you hoping for? How long will the drug last in the bloodstream before you need to re-administer it?
6. A dog with severe heart failure and low cardiac output is already being treated with digoxin, pimobendan, furosemide, enalapril and spironolactone. You decide that you want to start a continuous rate infusion of a catecholamine. What will you use?
How will you administer it? Can you calculate how much drug needs to be added to the fluids to achieve the correct CRI.
7. What is the mechanism of action for Pimobendan, and when might you use it?
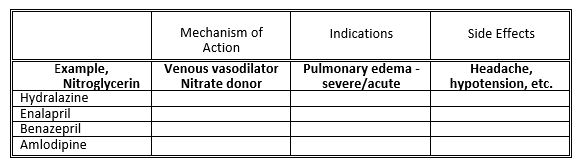
Vasodilators
1. Explain why an arterial vasodilator will help a patient with CHF.
2. Explain why a venous vasodilator will help a patient with CHF.
3. Characterize the side effects of excessive arterial or venous vasodilation.
4. Be familiar with the mechanism of action, the indications for and the side effects of the following drugs:
Summary of a possible therapy overview for management of heart disease:
Asymptomatic dog with heart disease
1. Avoid excessive salty foods
2. Avoid repetitive and strenuous exercise
3. Avoid obesity
4. ACE inhibitor? Beta-blocker? Pimobendan?
Early CHF in a dog
1. NO processed people food or salty treats
2. Avoid repetitive and strenuous exercise
3. Slowly transition to moderately sodium restricted diet, +/- omega-3 FA
4. ACE inhibitor and/or pimobendan
5. Low dose furosemide (2 mg/kg q 24 hrs)
6. Beta-blocker?
Moderate to severe CHF in a dog
1. NO processed people food or salty treats
2. Avoid significant exertion – leash walks only
3. Low to moderate sodium-restricted diet +/- omega-3 FA
4. ACE inhibitor – BID dose
5. Low dose furosemide (2-4 mg/kg q 12 hrs)
6. Pimobendan
7. +/-Beta-blocker if previously started and well tolerated
8. +/- Digoxin (esp. of atrial fibrillation or significant supraventricular arrhythmia)
9. +/- Spironolactone (+ hydrochlorothiazide)
10. ± amlodipine or hydralazine (esp. if significant mitral regurgitation with normal or high blood pressure)
Acute, fulminant CHF in a dog
1. No food needed, make sure free access to water
2. Cage rest – let out of cage to urinate
3. Furosemide 4 mg/kg IV q 1 hour until respirations improved, then q 6-12 hours
4. +/- Nitroglycerin transcutaneously
5. +/- furosemide CRI
6. If hypotensive and DCM, Dobutamine
7. If severe pulmonary edema, Nitroprusside
8. Once CHF improving then transition to oral furosemide, ACE inhibitor, pimobendan, or other cardiac medications.
ANGIOTENSIN CONVERTING ENZYME INHIBITORS
The angiotensin converting enzyme inhibitors are routinely used for management of CHF. Studies in humans and dogs demonstrate improved survival times (or time until decompensation for heart failure) in patients with advanced forms of heart failure. Additionally, human patients with preclinical heart failure may have prolonged asymptomatic periods after ACE inhibition, i.e., the development of heart failure may be postponed. The angiotensin converting enzyme (ACE) inhibitors lisinopril, benazepril, and enalapril are commonly used to treat CHF. ACE inhibitors act as balanced vasodilators, but they have other effects as well to alter cardiac remodeling and slow progressive cardiac enlargement. Most animals with moderate to severe heart failure have activation of the renin-angiotensin-aldosterone system. When this system is activated, angiotensin converting enzyme normally cleaves circulating angiotensin I to active angiotensin II. Angiotensin II is a potent vasoconstrictor that also stimulates release of aldosterone from the adrenal cortex. ACE inhibitors act as balanced vasodilators by abating the vasoconstrictive effects of angiotensin II on arteries and veins in addition to reducing the renal retention of sodium and water by secondarily inhibiting aldosterone release. These drugs are available in oral formulation, and enalapril also is available for IV injection. Side effects of ACE inhibitors include hypotension and deterioration of renal function, both of which can often be successfully relieved when the dose of diuretic is reduced. Enalapril can be started at 0.5 mg/kg q 24 h, but the target dose is 0.5 mg/kg q 12 h. These drugs have a fairly rapid onset of action (less than four hours) and can be useful in the initial management of acute congestive heart failure although the effects in this setting are modest compared to other drugs. ACE inhibitors are most useful for chronic treatment of CHF. ACE inhibitors are a mainstay of CHF therapy, and all dogs and most cats that can tolerate these drugs should be maintained on them.
Vasodilators
Vasodilators act by favorably altering the loading conditions of the heart such that forward cardiac output is improved or congestion is relieved. Certain vasodilators act as systemic venous dilators (venodilators) and cause preload reduction. Other vasodilators act on the systemic arterial tree (arterial dilators) and lead to arterial vasodilation, decreasing afterload and thereby improving cardiac performance. Some vasodilators cause both arterial and venous vasodilation (balanced vasodilators).
Arterial vasodilators are indicated in a variety of disease processes where afterload is felt to be elevated; generally most conditions associated with congestive heart failure except pericardial disease and stenotic valvular or subvalvular disease. Venous vasodilators are used in settings of excessive preload, especially in animals with severe pulmonary edema. Vasodilators commonly employed in veterinary medicine include amlodipine, nitroglycerin and hydralazine.
Nitroglycerin, the prototypic venodilator, causes direct relaxation of venous smooth muscle and increases systemic venous capacitance. This reduces preload and is most frequently used in animals for acute cardiogenic pulmonary edema. Nitroglycerin is usually used for 1-3 days in animals with pulmonary edema. Transcutaneous absorption of nitroglycerin paste facilitate drug administration, however non-permeable gloves should be worn during application and adhesive tape labeled “Nitrol” is placed on the outer surface of the ear to alert anyone working with the dog to avoid contact with the drug. Overdosage can lead to hypotension, depression, lethargy, nausea, and pre‑renal azotemia. The drug is applied to the inner surface of the pinna of the ear or other hairless area of the body (1/4″ to 1″ q 6 h – dogs, 1/8″ to 1/4″ q 6 h – cats). Nitroglycerin is typically used only until life threatening edema has resolved, as drug tolerance may develop.
Hydralazine or amlodipine are preferred by a few cardiologists as an add-on drug for animals with mitral valve insufficiency when standard heart failure drugs are no longer effective. Amlodipine is a calcium channel blocker with predominant effects on the arterial vasculature to reduce afterload and create a drop in blood pressure. Due to the seemingly reduced frequency of side effects for amlodipine compared to hydralazine, many cardiologists prefer amlodipine. Hydralazine is the classic direct acting arteriolar vasodilator. Hydralazine is rapidly absorbed from the intestinal tract and the onset of action occurs within 1 hour and peak improvement in cardiac performance occurring in 3 to 5 hours. The dose of hydralazine is titrated to between 1 to 3 mg/kg. Hydralazine is used either in the setting of severe pulmonary edema (when sodium nitroprusside is not practical) or in the setting of chronic mitral regurgitation, where the drug may result in decreased mitral regurgitant volume, with subsequent reduced left atrial size. Hypotension, anorexia, reflex sinus tachycardia and vomiting are reported complications.
In those animals with very severe pulmonary edema who do not respond initially to furosemide, IV sodium nitroprusside (Nipride, Hoffman‑La Roche) continuous infusion may prove useful. Sodium nitroprusside is a balanced vasodilator, acts directly on arterial and venous vascular smooth muscle. Intravenous administration of sodium nitroprusside by continuous infusion is required as the drug has a short duration of action. In the small number of canine patients with severe heart failure refractory to furosemide and other vasodilators, nitroprusside may be effective. Sodium nitroprusside is a potent hypotensive agent and should only be used where close monitoring of the arterial blood pressure, ECG, and patient is possible. The drug is susceptible to degradation in light and the prepared solution must be wrapped in the foil provided (read directions carefully). Initial dose of 1 mg/kg/minute is titrated toward 3 – 10 mg/kg/min.
Narcotics have both analgesic and anti-anxiety effects to help calm animals that are distressed and dyspneic, specifically dogs with CHF in which the stress of hospitalization leads to barking, pawing at the cage and excitability. Morphine may also slow the heart rate and act as a mild vasodilator. Butorphanol, oxymorphone, buprenorphine, or acepromazine may be substituted for morphine, although acepromazine should be avoided in animals that are hypotensive.
DIURETICS
Diuretics are at the foundation of congestive heart failure therapy. Diuretics are routinely used whenever heart disease is accompanied by evidence of congestion (pleural effusion, pulmonary edema, ascites). The pharmacologic mechanisms of action vary among the diuretic classes; however, all have a common action of inhibiting renal ion transport. The resulting loss of sodium and water results in preload reduction and improvement in signs of congestive heart failure. In accordance with the Frank Starling Law, excessive diuresis will result in signs of low output with weakness, dehydration and azotemia.
Loop or “High Ceiling” Diuretics
The Loop of Henle is the site of action of the most potent diuretics, which are also referred to as the “high-ceiling” diuretics. These diuretics can induce a natriuresis of approximately 20 to 25% of the filtered sodium load. Examples include furosemide (Lasix), torsemide and bumetanide. These drugs act to inhibit chloride and sodium resorption in the thick ascending Henle’s Loop by actively blocking the sodium-potassium-chloride cotransport. Furosemide has an extremely rapid onset of action, with diuresis ensuing within minutes and action peaking within 30 minutes. Cats may be more sensitive than dogs with respect to development of dehydration and azotemia following furosemide administration. Accordingly, a lower dose of furosemide is often used in cats as compared to dogs. Furosemide may be administered PO, IV, IM or subcutaneously. The canine dose varies from 1-4 mg/kg, once a day to TID. A continuous rate infusion of furosemide may be more effective in severe pulmonary edema and create fewer side effects that repeated bolus administration of the drug. Mild heart failure is treated with a low dose of furosemide; severe heart failure is treated with high doses. Side effects of furosemide may include hypokalemia, dehydration, azotemia and following rapid IV injection, deafness.
Potassium Sparing Diuretics
The potassium sparing diuretics are weak diuretic agents. These drugs may act through antagonism of aldosterone (spironolactone and eplerenone) and through direct inhibition of sodium transport (amiloride, triamterene). Aldosterone antagonists like spironolactone and eplerenone competitively bind to the set of plasmic receptor in aldosterone-responsive cells in the distal tubule and early collecting duct. By blocking potassium-sodium exchange they conserve potassium and promote sodium excretion. Through this action, they result in a mild diuresis. Perhaps more importantly, they conserve potassium which is typically lost with administration of other diuretic agents. Spironolactone may also have important aldosterone antagonist effects away from the heart, i.e. at the baroreceptors, antifibrotic effect in the myocardium. These drugs are usually used in combination with furosemide in animals with refractory CHF. They may be used after hypokalemia has proven to be a problem. Finally, recent human trials have indicated an increased survival when spironolactone or eplerenone were added to standard CHF therapies. Potassium sparing diuretics are usually contraindicated in animals with hyperkalemia, and concurrent administration of oral potassium supplements is no recommended.
Calcium Sensitizers and other Inotropic Agents – Pimobendan, Levosimendan, Amrinone and Milrinone
Pimobendan, levosimendan, and milrinone are oral vasodilator/ inotropes. Of these medications, only pimobendan is approved in veterinary medicine, although clinical trials have been completed looking into the utility of levosimendan for dogs with CHF. Amrinone and Milrinone are available for continuous infusion and can be employed in settings where Dobutamine might be used, animals with severe congestive heart failure and pulmonary edema. The effects of amrinone and milrinone result from inhibition of phosphodiesterase, specifically action on cardiac phosphodiesterase III. Proarrhythmia may be a problem with these drugs, however, it was not noted to be a significant problem in the veterinary clinical trials. Large trials in people, however, have proven that chronic administration of milrinone and amrinone lead to shortened survival (early death) in humans, and there are even some preliminary studies which suggest that acute administration of positive inotropes in an acute CHF setting might result in reduced survival compared to infusion of vasodilators.
Pimobendan has effects as both a calcium sensitizer and a cardiac phosphodiesterase inhibitor. Pimobendan is available in an oral formulation, and several veterinary studies have documented the efficacy of pimobendan and in dogs with dilated cardiomyopathy and chronic valvular disease. Pimobendan is approved for use in dogs with DCM or chronic mitral valve disease AFTER the onset of CHF. The drug has been shown to be at least equivalent to and perhaps superior to ACE inhibitors in the management of CHF in dogs. Pimobendan also seems to have a good safety margin and side effects are infrequent. Levosimendan is also a calcium sensitizer, and has vasodilator actions via opening ATP-sensitive potassium channels in smooth muscle.
ADRENERGIC – RECEPTOR ACTIVITY OF SYMPATHOMIMETIC AMINES
The sympathomimetic amines can be used to stimulate a and ß receptors and result in increased vascular tone, increased inotropy, increased heart rate, increased cardiac conduction and in some instances, vasodilation and bronchodilation. Stimulation of a receptors leads to vasoconstriction. There are some a receptors on the heart that also increase inotropic state. By far and away, however, the majority of the adrenergic receptors in the heart are ß1 receptors. Stimulation of ß1 receptors leads to increased heart rate, increase inotropic state and arrhythmogenicity. ß2 receptors lie on blood vessels and bronchi and stimulation of ß2 receptors leads to vasodilation and bronchodilation. Dopaminergic receptors lie on renal, mesenteric, cerebral and coronary arterial beds and stimulation of these receptor leads to local vasodilatation in these tissues.
In the management of chronic congestive heart failure, increases in afterload caused by alpha-adrenergic stimulation provide additional stress to the heart and oppose ventricular ejection. For this reason, norepinephrine and epinephrine are rarely used in a setting of chronic congestive heart failure. Dopamine may be useful at mid-range doses for acute congestive heart failure. The three dose ranges for dopamine are 1-3 mg/kg/minute for stimulation of dopaminergic receptors in the renal beds leading to vasodilation. The mid-range dose is 5-10 mg/kg/minute and can be used to support cardiac function. At higher doses (10-40 mg/kg/minute) dopamine causes a stimulation and leads to significant increases in blood pressure. Therefore, these high doses are not used in the setting of congestive heart failure and are more commonly used in systemic shock without congestion. Isoproterenol, the final adrenergic listed in the table below, has no a activity and a large ß2 activity which results in vasodilation. Unopposed ß2 stimulation may lead to excessive hypotension and inadequate organ perfusion.
All sympathomimetic amines have short half-lives, along the lines of minutes, and, therefore, must be given by continuous rate infusion to achieve continuous blood levels and effects. Close monitoring is required for the use of these medications. Such monitoring includes continuous ECG monitoring, intermittent blood pressure monitoring and attentive nursing care. In practice, congestive heart failure is usually treated with dobutamine in a dose range of 2.5-10 mg/kg/minute. After two to three days, down regulation of adrenergic receptors makes these drugs less effective. For this reason, dobutamine is typically used for a 2-3 day infusion until other cardiac medications have taken effect.
Hemodynamic effects of ideal sympathomimetic drug
1. Increase cardiac output (CO), stroke volume (SV) and improve blood flow to all organs; improve contractility and maintain arterial blood pressure.
2. Have little or no effect upon cardiac rate and rhythm; non-arrhythmogenic.
3. Improve ratio of myocardial oxygen demand to myocardial oxygen supply.
Potential Detrimental effects of catechols
1. Hypotension (isoproterenol)
2. Hypertension (NE, dopamine, epinephrine)
3. Cardiac arrhythmias (may be seen with all adrenergic drugs)
4. Increased myocardial oxygen consumption

REFERENCES
1. Current Veterinary Therapy XIV, Bonagura JD, Twedt DC (eds) Saunders 2009
Keene BW, Bonagura JD. Management of Heart Failure in dogs
Rush JE. Chronic Valvular Disease in dogs
Hogan DF, Green HW. Dilated cardiomyopathy in Dogs
Luis Fuentes V. Management of Feline Myocardial Disease.
2. Textbook of Veterinary Internal Medicine. Ettinger SJ, Feldman EC (eds). Elsevier Saunders 2010 Bulmer BJ, Sisson DD. Therapy of Heart Failure