Projects Overview
- Nanoparticle and clean water: Titanium Ultranano Particle
- Substrate for catalysts: Silica scaffold
- Ice: single crystal, nucleation, and anti-icing
- Environment: ammonia-air interface
- Instrument development: SBW-M Nonlinear Interferometer
Nanoparticle for clean water
The clean water project focuses on eliminating organic contaminants, bacteria, and pharmaceutical residues from drinking water. According to the United Nations, four out of ten people face water scarcity, and exposure to polluted water—especially long-lasting pollutants like POPs—can pose serious health risks and even be deadly.
This project builds on the remarkable oxidation potential of titanium dioxide (TiO2)—the same compound responsible for the white color in toothpaste, skim milk, and salad dressing. TiO2 is inert, biocompatible, and widely employed in wastewater treatment processes, yet it suffers from critical limitations. Despite being one of the most extensively studied photocatalysts, the efficiency of commercial TiO2 is hindered by its wide bandgap, rapid electron–hole recombination, and extremely low solubility in water, all of which restrict its direct applicability in aqueous environments.
To address these challenges, the Shultz group engineered ultrasmall anatase TiO2 particles (less than two nanometers in diameter), termed TiUNP. These innovations increased efficiency by nearly an order of magnitude and enabled the particles to effectively purify drinking water from persistent organic pollutants (POPs). This research is supported by the U.S. Department of Energy (DE-SC0020258).

Lin et al. 2024
TiUNPs can be doped with different single transition-metal atoms to tune its behavior. To date, we have developed the capability to incorporate the full spectrum of 4th-row transition metal ions into TiUNPs, enabling precise tailoring of photocatalyst properties for a wide range of applications.

Lin et al. 2024
Unlike conventional Fenton-style photocatalysts, Fe•TiUNP utilize a direct charge transfer (DCT) pathway, bypassing short-lived radical intermediates. This unique mechanism enables the particles to oxidize organic pollutants in a more efficient, predictable and controlled manner.

Lin et al. 2025
Substrate for catalysts
When working with nanoscale catalysts, a dispersive environment is essential to maximize catalytic efficiency. Owing to their hydrophilic nature and ultra-small size, TiUNPs readily form homogeneous solutions in water. However, separating and recovering such small particles presents a significant challenge. To address this, our current approach focuses on immobilizing the particles onto silica substrates or scaffolds.



Conventional scaffolds typically resemble a loose mesh, providing support but leaving large, irregular gaps. In contrast, our design mimics a tightly bundled arrangement of straws, forming a true honeycomb-like structure. This orderly architecture not only offers greater stability but also establishes a more efficient framework, allowing water to flow through more freely and consistently.

Light transmittance is a critical parameter for any substrate intended to support a photocatalyst. Our material behaves much like an optical fiber, guiding and transmitting light efficiently through its structure. With a wide band gap of about 7 eV, scaffold does not interfere with the activity of TiUNP, whose band gap is around 3.2 eV.
Ice: single crystal, nucleation, and anti-icing
Prof. Shultz has developed a technique to grow high-quality single crystal ice based on a modified Bridgeman apparatus. Targeted ice faces can be cut from the ice boules based on the orientation of the optical axis of the crystal. Being able to produce high-quality single crystals is necessary for obtaining reliable SFG spectra of different ice faces and is useful for future research involving ice.

The Shultz lab participates in a multidisciplinary university research initiative (MURI) on ice nucleation and anti-icing, collaborating with theoretical and experimental groups. Current studies focus on the ice recrystallization inhibitor poly(vinyl alcohol) (PVA).


Environment: ammonia
The current environmental project focuses on ammonia. The ability to turn molecular nitrogen into ammonia (fertilizer) has been characterized as the single most important chemical discovery ever: it is responsible for half the nitrogen atoms in every person’s proteins! Unfortunately, half of all fertilizer escapes, polluting water (think bright green algae) or finds its way into the atmosphere. Ammonia’s fate in the atmosphere is largely unknown. What is known is that ammonia is the only significant concentration basic gas in the atmosphere. As a base, it reacts with the plethora of acidic gases to form solids known as cloud condensation nuclei. Clouds, in turn, are the largest uncertainty for understanding the environment. This project focuses on interaction between aqueous solutions and ice with ammonia. The ultimate goal from understanding these interactions is to design fertilizers that hold ammonia in the soil to be accessible to the plants it was put there to fertilize. Current opportunities focus on characterizing the fundamental interactions, supported by NSF Award # 1807913


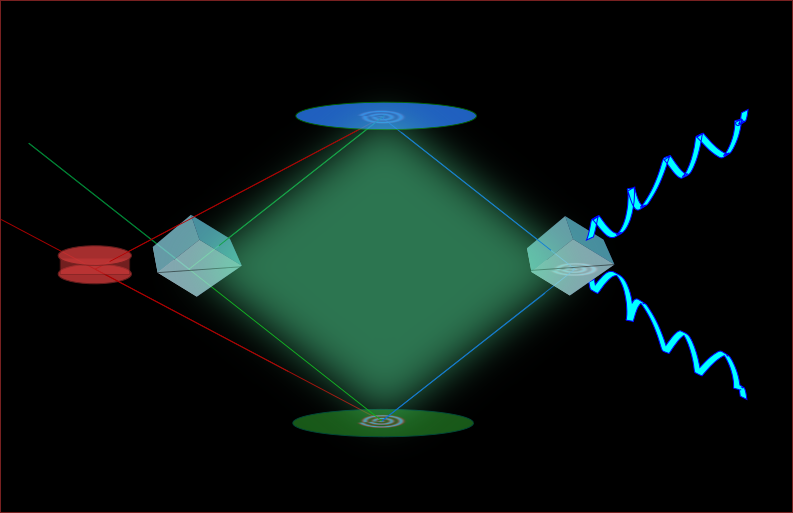
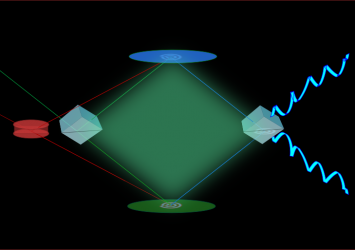
Instrument development
Whether developing the photocatalyst scaffold or investigating ammonia-solution Whether developing the photocatalyst scaffold or investigating ammonia-solution interactions, we require suitable instrumentation. Although numerous tools exist for probing solid – especially metal – surfaces, tools for probing complex, flexible, high vapor pressure interfaces are just being developed. The Shultz group invented one: a nonlinear interferometer. Since any surface contains seven to ten orders of magnitude fewer molecules than the bulk phases on either side of the interface, the first requirement is great sensitivity to the surface. For complex mixtures, the second requirement is being able to distinguish among the multiple surface species. The Shultz group uses a technique called sum frequency generation (SFG) to provide the surface sensitivity. Distinguishing among multiple surface species and measuring their concentration requires measuring a complex (in the mathematical sense) signal. We invented a nonlinear interferometer to measure the complex signal. Our current target is to extend this instrument to measure small differences as the surface molecules react to generate new materials. Materials that interact on ice surfaces likely change the surface morphology – either roughening or leveling it. The target of the not-yet-existing Nomariscope is to detect the morphology changes. We are now having three nonlinear systems for different research needs:1) HAL system, which has successfully measured solid surfaces. 2)Diana system, which has successfully measured solid and about to realize the expansion to liquid sample. 3)Zeus system, an advanced femto-second system which is under construction. Opportunities exist to get involved with creating instruments that enable discoveries well beyond the Shultz laboratory.