Cytochrome P450 2E1
With increasing blood alcohol concentration, a secondary pathway for ethanol metabolism kicks in using the microsomal cytochrome P450 enzyme CYP2E1 (7). When the ethanol concentration is low, CYP2E1 is only responsible for oxidizing around 10% of the ethanol, but as the blood alcohol concentration increases, so does the activity of CYP2E1 in metabolizing ethanol. Chronic alcohol consumption increases the CYP2E1 levels, which allow the enzyme to play a larger role in ethanol metabolism in chronic alcoholics. In addition to the oxidation of ethanol, CYP2E1 also oxidizes a variety of substrates including certain drugs (3).
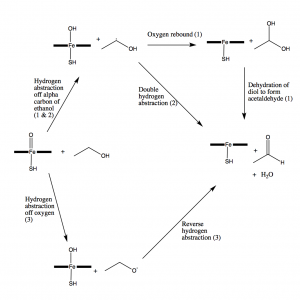
Figure 8: Three proposed pathways for the oxidation of ethanol to acetaldehyde by cytochrome P450 2E1.
There are three proposed mechanisms for ethanol oxidation by CYP2E1, all utilizing radical chemistry, as shown in the scheme above. The first mechanism is initiated by a hydrogen abstraction from the alpha carbon of ethanol, followed by the rebound of oxygen, forming a gem-diol intermediate which is then dehydrated to produce acetaldehyde and regenerate the gem-diol. The second mechanism is characterized by a dual-hydrogen abstraction, in which the first hydrogen is abstracted from the alpha carbon of ethanol, and then another hydrogen is abstracted from the oxygen in the hydroxyl group of ethanoyl radical, bypassing the formation of the gem-diol intermediate and directly producing acetaldehyde (8). These two mechanisms beginning with the hydrogen abstraction from the alpha carbon of ethanol are proposed in multiple papers throughout the past 20 years, thus they are heavily supported in the primary literature (9). After discovering that the CYP2E1 active site is sterically unhindered above the iron center, Wang et al. proposed a new, third mechanism which they appropriately named a reversed dual-hydrogen abstraction. In this mechanism, the first hydrogen is abstracted from the oxygen of ethanol, followed by a second hydrogen abstraction from the alpha carbon of ethanol to produce acetaldehyde (8).
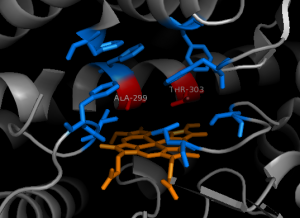
Figure 9: The active site of CYP2E1. Ala299 and Thr303 function as the active residues, while the blue residues form the binding pocket (pbd file: 3e6i).
Wang et al. determined that the reverse dual-hydrogen abstraction (R-DHA) mechanism is predominant in polar environments, while in nonpolar environments the oxidation of ethanol is carried out competitively through the gem-diol mechanism and the R-DHA mechanism. This conclusion led Wang et al. to propose an argument for how CYP2E1 is able to adapt a significant role in ethanol oxidation when the blood ethanol concentration is high. The discovery that ethanol is a modulator of CYP2E1 substrate metabolism, signifying that ethanol oxidation by CYP2E1 can be induced by ethanol, was the key event that allowed Wang et al. to be able to explain this adaptive role of CYP2E1 (8).
When blood ethanol concentration is low, there is only a small amount of ethanol in the heme pocket, contributing to a small bulk polarity which causes the competition between the gem-diol and R-DHA mechanisms to occur. When blood ethanol concentration rises, more and more ethanol molecules enter the heme pocket, increasing the bulk polarity, allowing the R-DHA mechanism to become the primary mechanism through which ethanol oxidation occurs which in turn causes the rate of ethanol metabolism by CYP2E1 to increase as the competitive factor is eliminated (8).
November 7, 2016 at 7:32 pm
Could you make your figures a little bit larger? On my screen they show up quite small and the small text is a little difficult to read.
I think Figure 11 is a good view of the active site. You say Ala299 and Thr303 are the active residues; could you explain somewhere how?
November 10, 2016 at 4:20 pm
The text is pretty small in Figure 10, enlarging that figure might be beneficial.
On your Answer page, I mentioned wanting differences between cytochrome P450, catalase, and ADH, which you have shown me here, perhaps rendering the inclusion of more details about their differences on the Answer page unnecessary. However, the issue with alcohol’s effect in conjunction with other drugs remains, as in the end of the first paragraph here you briefly mention that CYP2E1 can also oxidize a variety of substrates including certain drugs. This is a cool fact, and should remain on this page, but it does manifest some discontinuity between this page and the Answer page.
November 10, 2016 at 4:23 pm
I guess the discontinuity is really between the question, answer and this page. Not just this page and the answer page. If the question page didn’t include that second question, there wouldn’t be any discontinuity.
August 2, 2023 at 4:50 am
egfzrxzr http://para-mayores.es/# ibazvnyc
August 7, 2023 at 4:26 am
accutane costs https://isotretinoinacne.shop/# buy accutane canada pharmacy
August 20, 2023 at 7:59 am
similar to cialis tadalafilise.cyou/#
August 22, 2023 at 3:40 am
Hello there, simply was alert to your blog thru Google, and located that it is really informative. I?m going to be careful for brussels. I will be grateful in the event you proceed this in future. Many people can be benefited out of your writing. Cheers!
August 23, 2023 at 5:08 am
cialis 60mg for sale best price tadalafil online cialis daily use coupon
August 23, 2023 at 6:51 pm
What?s Happening i am new to this, I stumbled upon this I have discovered It absolutely helpful and it has aided me out loads. I am hoping to give a contribution & help different customers like its helped me. Great job.
August 24, 2023 at 4:16 pm
I have been surfing online more than 3 hours lately, yet I never found any attention-grabbing article like yours. It is lovely worth sufficient for me. Personally, if all webmasters and bloggers made excellent content as you did, the net will likely be a lot more useful than ever before.
August 25, 2023 at 12:46 am
Pretty great post. I simply stumbled upon your blog and wanted to say that I have really enjoyed browsing your weblog posts. After all I?ll be subscribing for your rss feed and I hope you write again soon!
August 25, 2023 at 6:50 am
200 mg cialis free sample pack of cialis best over the counter viagra substitute
August 26, 2023 at 12:50 am
Hi there I am so thrilled I found your site, I really found you by mistake, while I was searching on Google for something else, Regardless I am here now and would just like to say cheers for a marvelous post and a all round thrilling blog (I also love the theme/design), I don’t have time to read it all at the minute but I have bookmarked it and also included your RSS feeds, so when I have time I will be back to read a great deal more, Please do keep up the superb job.
August 27, 2023 at 2:15 am
cialis in malaysia cialis 20 mg in india generic for cialis tadalafil
August 27, 2023 at 4:30 am
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You obviously know what youre talking about, why throw away your intelligence on just posting videos to your site when you could be giving us something enlightening to read?
August 27, 2023 at 2:28 pm
Heya i am for the primary time here. I came across this board and I find It truly helpful & it helped me out much. I am hoping to offer one thing back and aid others such as you helped me.
August 27, 2023 at 6:17 pm
very nice post, i definitely love this web site, keep on it
August 28, 2023 at 7:40 am
One thing I have actually noticed is that there are plenty of fallacies regarding the banks intentions if talking about foreclosures. One fantasy in particular is always that the bank desires your house. The lending company wants your dollars, not your house. They want the funds they gave you along with interest. Averting the bank will only draw a foreclosed realization. Thanks for your post.
August 28, 2023 at 10:24 pm
cialis for bph covered by medicare cialis usa pharmacy daily cialis
August 29, 2023 at 2:56 am
drugs from canada https://certifiedcanadapills.pro/# reddit canadian pharmacy
August 29, 2023 at 7:03 am
fantastic post, very informative. I wonder why the other experts of this sector do not notice this. You must continue your writing. I’m confident, you have a great readers’ base already!
August 29, 2023 at 8:00 am
best canadian online pharmacy canadian pharmacy ed medications canadian pharmacy meds reviews
August 29, 2023 at 8:17 am
best india pharmacy: buy prescription drugs from india – online pharmacy india
August 29, 2023 at 8:18 am
pharmacies in mexico that ship to usa medicine in mexico pharmacies purple pharmacy mexico price list
August 29, 2023 at 8:22 am
http://mexicanpharmacy.guru/# mexican online pharmacies prescription drugs
August 29, 2023 at 8:27 am
buying from canadian pharmacies https://certifiedcanadapills.pro/# online canadian drugstore
August 29, 2023 at 8:36 am
mexico pharmacies prescription drugs medication from mexico pharmacy buying from online mexican pharmacy
August 29, 2023 at 8:50 am
canada pharmacy online: legit canadian pharmacy – canadian pharmacy meds
August 29, 2023 at 8:54 am
online shopping pharmacy india top 10 online pharmacy in india indian pharmacy paypal
August 29, 2023 at 8:55 am
reputable canadian online pharmacy: ed drugs online from canada – canada drugstore pharmacy rx
August 29, 2023 at 8:58 am
medication canadian pharmacy https://certifiedcanadapills.pro/# reliable canadian pharmacy reviews
August 29, 2023 at 8:58 am
http://indiapharmacy.cheap/# india online pharmacy
August 29, 2023 at 9:08 am
canadian pharmacy no rx needed: my canadian pharmacy rx – online canadian pharmacy reviews
August 29, 2023 at 9:12 am
pharmacies in mexico that ship to usa mexican pharmaceuticals online mexico drug stores pharmacies
August 29, 2023 at 9:16 am
http://mexicanpharmacy.guru/# purple pharmacy mexico price list
August 29, 2023 at 9:25 am
india pharmacy: india pharmacy – п»їlegitimate online pharmacies india
August 29, 2023 at 9:28 am
canadian pharmacy 365 http://certifiedcanadapills.pro/# reputable canadian pharmacy
August 29, 2023 at 9:30 am
reputable mexican pharmacies online medication from mexico pharmacy buying prescription drugs in mexico
August 29, 2023 at 9:33 am
https://certifiedcanadapills.pro/# reliable canadian pharmacy
August 29, 2023 at 9:43 am
canada rx pharmacy: canada drug pharmacy – canadian pharmacy near me
August 29, 2023 at 9:48 am
buying from online mexican pharmacy purple pharmacy mexico price list buying from online mexican pharmacy
August 29, 2023 at 9:51 am
https://mexicanpharmacy.guru/# mexico pharmacies prescription drugs
August 29, 2023 at 9:59 am
northwest canadian pharmacy https://certifiedcanadapills.pro/# canadian pharmacies online
August 29, 2023 at 10:00 am
cheap canadian pharmacy online: trusted canadian pharmacy – canadian pharmacy reviews
August 29, 2023 at 10:05 am
canadianpharmacymeds my canadian pharmacy canadian world pharmacy
August 29, 2023 at 10:08 am
https://certifiedcanadapills.pro/# canadian drug pharmacy
August 29, 2023 at 10:11 am
india pharmacy: india online pharmacy – reputable indian online pharmacy
August 29, 2023 at 10:18 am
pharmacy website india: п»їlegitimate online pharmacies india – cheapest online pharmacy india
August 29, 2023 at 10:25 am
https://mexicanpharmacy.guru/# buying prescription drugs in mexico
August 29, 2023 at 10:29 am
online canadian drugstore https://certifiedcanadapills.pro/# my canadian pharmacy
August 29, 2023 at 10:41 am
canadian pharmacy phone number canadian discount pharmacy northern pharmacy canada
August 29, 2023 at 10:43 am
https://indiapharmacy.cheap/# indian pharmacy paypal
August 29, 2023 at 10:49 am
medication from mexico pharmacy: п»їbest mexican online pharmacies – mexican mail order pharmacies
August 29, 2023 at 10:53 am
buying from online mexican pharmacy: medicine in mexico pharmacies – п»їbest mexican online pharmacies
August 29, 2023 at 11:00 am
legit canadian pharmacy online http://certifiedcanadapills.pro/# canadian discount pharmacy
August 29, 2023 at 11:11 am
india pharmacy: buy prescription drugs from india – canadian pharmacy india
August 29, 2023 at 11:19 am
mexico pharmacies prescription drugs mexican drugstore online п»їbest mexican online pharmacies
August 29, 2023 at 11:27 am
mexican border pharmacies shipping to usa: reputable mexican pharmacies online – mexican border pharmacies shipping to usa
August 29, 2023 at 11:29 am
buying prescription drugs in mexico online: mexico drug stores pharmacies – mexican border pharmacies shipping to usa
August 29, 2023 at 11:31 am
canadian pharmacy https://certifiedcanadapills.pro/# canadian valley pharmacy
August 29, 2023 at 11:38 am
mexican online pharmacies prescription drugs mexico pharmacies prescription drugs buying prescription drugs in mexico
August 29, 2023 at 11:39 am
http://certifiedcanadapills.pro/# canadian pharmacy no scripts
August 29, 2023 at 11:47 am
п»їbest mexican online pharmacies: mexico drug stores pharmacies – best online pharmacies in mexico
August 29, 2023 at 11:57 am
prescription drugs canada buy online canadian drugs online canadianpharmacyworld
August 29, 2023 at 11:57 am
http://indiapharmacy.cheap/# world pharmacy india
August 29, 2023 at 12:01 pm
safe reliable canadian pharmacy https://certifiedcanadapills.pro/# canadian family pharmacy
August 29, 2023 at 12:05 pm
Online medicine home delivery: online shopping pharmacy india – buy medicines online in india
August 29, 2023 at 12:15 pm
https://mexicanpharmacy.guru/# buying prescription drugs in mexico online
August 29, 2023 at 12:16 pm
vipps approved canadian online pharmacy canadian world pharmacy cheap canadian pharmacy online
August 29, 2023 at 12:23 pm
india pharmacy mail order: reputable indian pharmacies – indian pharmacy
August 29, 2023 at 12:32 pm
safe canadian pharmacy http://certifiedcanadapills.pro/# www canadianonlinepharmacy
August 29, 2023 at 12:33 pm
https://certifiedcanadapills.pro/# canadian online drugstore
August 29, 2023 at 12:33 pm
mexico drug stores pharmacies п»їbest mexican online pharmacies mexican drugstore online
August 29, 2023 at 12:40 pm
mexico pharmacies prescription drugs: mexico drug stores pharmacies – mexican drugstore online
August 29, 2023 at 12:44 pm
mexico pharmacies prescription drugs: medicine in mexico pharmacies – purple pharmacy mexico price list
August 29, 2023 at 12:50 pm
pharmacy website india top online pharmacy india Online medicine order
August 29, 2023 at 12:58 pm
mexican mail order pharmacies: mexico drug stores pharmacies – mexican drugstore online
August 29, 2023 at 1:03 pm
canada pharmacy http://certifiedcanadapills.pro/# canadian pharmacy world reviews
August 29, 2023 at 1:07 pm
https://certifiedcanadapills.pro/# canadian pharmacy ltd
August 29, 2023 at 1:08 pm
best online pharmacy india indian pharmacy online indian pharmacy
August 29, 2023 at 1:16 pm
reputable canadian pharmacy: canadian pharmacy india – canadian medications
August 29, 2023 at 1:22 pm
pharmacies in mexico that ship to usa: pharmacies in mexico that ship to usa – mexican drugstore online
August 29, 2023 at 1:27 pm
best india pharmacy top 10 online pharmacy in india п»їlegitimate online pharmacies india
August 29, 2023 at 1:34 pm
legit canadian pharmacy http://certifiedcanadapills.pro/# canadian drugs pharmacy
August 29, 2023 at 1:44 pm
https://mexicanpharmacy.guru/# mexican border pharmacies shipping to usa
August 29, 2023 at 1:46 pm
mexico drug stores pharmacies best online pharmacies in mexico pharmacies in mexico that ship to usa
August 29, 2023 at 1:55 pm
п»їbest mexican online pharmacies: mexican rx online – п»їbest mexican online pharmacies
August 29, 2023 at 2:02 pm
best rated canadian pharmacy: safe online pharmacies in canada – reputable canadian pharmacy
August 29, 2023 at 2:03 pm
https://certifiedcanadapills.pro/# legit canadian pharmacy online
August 29, 2023 at 2:05 pm
Online medicine home delivery india online pharmacy Online medicine order
August 29, 2023 at 2:07 pm
canadian pharmacy near me https://certifiedcanadapills.pro/# canadian pharmacy store
August 29, 2023 at 2:14 pm
medicine in mexico pharmacies: buying prescription drugs in mexico online – п»їbest mexican online pharmacies
August 29, 2023 at 2:22 pm
https://indiapharmacy.cheap/# indian pharmacy online
August 29, 2023 at 2:33 pm
medication from mexico pharmacy: mexican pharmaceuticals online – mexican drugstore online
August 29, 2023 at 2:39 pm
ed meds online canada https://certifiedcanadapills.pro/# trustworthy canadian pharmacy
August 29, 2023 at 2:41 pm
http://mexicanpharmacy.guru/# п»їbest mexican online pharmacies
August 29, 2023 at 2:42 pm
mexico drug stores pharmacies: mexican pharmaceuticals online – buying prescription drugs in mexico online
August 29, 2023 at 2:45 pm
mexico drug stores pharmacies pharmacies in mexico that ship to usa reputable mexican pharmacies online
August 29, 2023 at 2:53 pm
pharmacy website india: mail order pharmacy india – indian pharmacy online
August 29, 2023 at 3:00 pm
http://mexicanpharmacy.guru/# buying prescription drugs in mexico
August 29, 2023 at 3:04 pm
indian pharmacy top 10 online pharmacy in india п»їlegitimate online pharmacies india
August 29, 2023 at 3:11 pm
canadian pharmacy meds review http://certifiedcanadapills.pro/# thecanadianpharmacy
August 29, 2023 at 3:12 pm
mexican border pharmacies shipping to usa: mexican rx online – purple pharmacy mexico price list
August 29, 2023 at 3:19 pm
http://certifiedcanadapills.pro/# real canadian pharmacy
August 29, 2023 at 3:21 pm
Online medicine order: best india pharmacy – indian pharmacy paypal
August 29, 2023 at 3:31 pm
canadian pharmacy 365: trustworthy canadian pharmacy – vipps canadian pharmacy
August 29, 2023 at 3:38 pm
https://mexicanpharmacy.guru/# buying from online mexican pharmacy
August 29, 2023 at 3:43 pm
best canadian pharmacy online https://certifiedcanadapills.pro/# canadian pharmacy 24
August 29, 2023 at 3:44 pm
mexican mail order pharmacies mexican mail order pharmacies mexican online pharmacies prescription drugs
August 29, 2023 at 3:58 pm
https://mexicanpharmacy.guru/# medicine in mexico pharmacies
August 29, 2023 at 4:00 pm
pharmacies in mexico that ship to usa: reputable mexican pharmacies online – mexican border pharmacies shipping to usa
August 29, 2023 at 4:04 pm
pharmacies in mexico that ship to usa mexican pharmaceuticals online mexican mail order pharmacies
August 29, 2023 at 4:10 pm
online canadian pharmacy review: canadian drugs online – pharmacy canadian superstore
August 29, 2023 at 4:15 pm
canadian mail order pharmacy http://certifiedcanadapills.pro/# my canadian pharmacy rx
August 29, 2023 at 4:17 pm
http://indiapharmacy.cheap/# Online medicine home delivery
August 29, 2023 at 4:24 pm
buying prescription drugs in mexico buying from online mexican pharmacy mexican drugstore online
August 29, 2023 at 4:38 pm
http://certifiedcanadapills.pro/# canadian pharmacy meds review
August 29, 2023 at 4:40 pm
purple pharmacy mexico price list: п»їbest mexican online pharmacies – mexican border pharmacies shipping to usa
August 29, 2023 at 4:46 pm
reputable mexican pharmacies online pharmacies in mexico that ship to usa mexican border pharmacies shipping to usa
August 29, 2023 at 4:48 pm
trusted canadian pharmacy https://certifiedcanadapills.pro/# canadian pharmacy meds review
August 29, 2023 at 4:51 pm
buy medicines online in india: indianpharmacy com – india online pharmacy
August 29, 2023 at 4:58 pm
https://indiapharmacy.cheap/# indian pharmacies safe
August 29, 2023 at 5:04 pm
advertorialpromovare.ro
August 29, 2023 at 5:06 pm
reputable indian online pharmacy pharmacy website india indian pharmacy online
August 29, 2023 at 5:18 pm
https://indiapharmacy.cheap/# cheapest online pharmacy india
August 29, 2023 at 5:20 pm
pet meds without vet prescription canada: precription drugs from canada – canadian pharmacy ltd
August 29, 2023 at 5:20 pm
northwest pharmacy canada https://certifiedcanadapills.pro/# canadian pharmacy king reviews
August 29, 2023 at 5:25 pm
top online pharmacy india п»їlegitimate online pharmacies india india online pharmacy
August 29, 2023 at 5:31 pm
buying from online mexican pharmacy: reputable mexican pharmacies online – mexican drugstore online
August 29, 2023 at 5:37 pm
https://certifiedcanadapills.pro/# reliable canadian pharmacy
August 29, 2023 at 5:44 pm
mexico drug stores pharmacies mexico drug stores pharmacies mexico pharmacies prescription drugs
August 29, 2023 at 5:50 pm
top online pharmacy india: top 10 online pharmacy in india – top online pharmacy india
August 29, 2023 at 5:52 pm
northwest canadian pharmacy http://certifiedcanadapills.pro/# canadian family pharmacy
August 29, 2023 at 5:56 pm
https://certifiedcanadapills.pro/# canada rx pharmacy
August 29, 2023 at 6:04 pm
mexican mail order pharmacies mexico drug stores pharmacies medicine in mexico pharmacies
August 29, 2023 at 6:15 pm
http://mexicanpharmacy.guru/# mexican pharmaceuticals online
August 29, 2023 at 6:23 pm
canada pharmacy online http://certifiedcanadapills.pro/# reliable canadian online pharmacy
August 29, 2023 at 6:24 pm
india online pharmacy buy medicines online in india cheapest online pharmacy india
August 29, 2023 at 6:34 pm
http://certifiedcanadapills.pro/# best online canadian pharmacy
August 29, 2023 at 6:38 pm
buy prescription drugs from india: buy medicines online in india – top online pharmacy india
August 29, 2023 at 6:44 pm
mexican border pharmacies shipping to usa mexican rx online mexican border pharmacies shipping to usa
August 29, 2023 at 6:48 pm
pharmacies in canada that ship to the us: canadian pharmacy 365 – canada pharmacy online legit
August 29, 2023 at 6:54 pm
http://indiapharmacy.cheap/# india online pharmacy
August 29, 2023 at 6:55 pm
adderall canadian pharmacy http://certifiedcanadapills.pro/# canada drugs reviews
August 29, 2023 at 7:04 pm
canadian pharmacy 365 reputable canadian pharmacy canadian medications
August 29, 2023 at 7:13 pm
http://indiapharmacy.cheap/# indian pharmacies safe
August 29, 2023 at 7:18 pm
mexican online pharmacies prescription drugs: mexican rx online – best online pharmacies in mexico
August 29, 2023 at 7:24 pm
buying from online mexican pharmacy buying prescription drugs in mexico mexican rx online
August 29, 2023 at 7:27 pm
northern pharmacy canada http://certifiedcanadapills.pro/# canadian pharmacy oxycodone
August 29, 2023 at 7:32 pm
http://mexicanpharmacy.guru/# п»їbest mexican online pharmacies
August 29, 2023 at 7:44 pm
mexico drug stores pharmacies п»їbest mexican online pharmacies reputable mexican pharmacies online
August 29, 2023 at 7:51 pm
http://certifiedcanadapills.pro/# the canadian pharmacy
August 29, 2023 at 7:58 pm
best canadian online pharmacy https://certifiedcanadapills.pro/# canadian pharmacy in canada
August 29, 2023 at 8:05 pm
online canadian pharmacy reviews: canadadrugpharmacy com – canadian pharmacy victoza
August 29, 2023 at 8:05 pm
reputable indian pharmacies indian pharmacy paypal pharmacy website india
August 29, 2023 at 8:13 pm
http://certifiedcanadapills.pro/# canadian medications
August 29, 2023 at 8:20 pm
canada drugs online https://certifiedcanadapills.pro/# best canadian online pharmacy
August 29, 2023 at 8:24 pm
best online pharmacy india: india online pharmacy – top online pharmacy india
August 29, 2023 at 8:44 pm
pharmacy website india: cheapest online pharmacy india – india online pharmacy
August 30, 2023 at 8:39 am
bogdan sirbu
August 30, 2023 at 10:07 am
Great write-up, I?m regular visitor of one?s blog, maintain up the nice operate, and It’s going to be a regular visitor for a lengthy time.
August 31, 2023 at 2:10 am
I haven?t checked in here for a while since I thought it was getting boring, but the last several posts are great quality so I guess I?ll add you back to my everyday bloglist. You deserve it my friend 🙂
August 31, 2023 at 4:07 am
http://edpill.men/# natural ed medications
August 31, 2023 at 4:08 am
order kamagra oral jelly: buy kamagra – order kamagra oral jelly
August 31, 2023 at 4:10 am
erection pills: ed meds online – what are ed drugs
August 31, 2023 at 4:18 am
Kamagra tablets: Kamagra tablets – buy kamagra online
August 31, 2023 at 4:20 am
natural ed medications treatment of ed generic ed drugs
August 31, 2023 at 4:28 am
Kamagra tablets 100mg: cheap kamagra – Kamagra tablets 100mg
August 31, 2023 at 4:31 am
order kamagra oral jelly: kamagra oral jelly – order kamagra oral jelly
August 31, 2023 at 4:36 am
http://kamagra.men/# order kamagra oral jelly
August 31, 2023 at 4:49 am
male ed drugs: ed pills for sale – best ed medications
August 31, 2023 at 4:51 am
kamagra oral jelly: order kamagra oral jelly – buy kamagra
August 31, 2023 at 5:01 am
https://cialis.science/# generic cialis paypal payment
August 31, 2023 at 5:03 am
kamagra Kamagra tablets Kamagra tablets 100mg
August 31, 2023 at 5:06 am
best ed drugs: erection pills – cheap erectile dysfunction pills online
August 31, 2023 at 5:10 am
cialis 20 mg sale: Buy Cialis online – cialis prices walmart
August 31, 2023 at 5:12 am
cialis super active cheap: Buy Tadalafil 20mg – blood pressure on cialis
August 31, 2023 at 5:26 am
https://cialis.science/# order cialis online us
August 31, 2023 at 5:31 am
online ed pills: cheap erectile dysfunction pills – medications for ed
August 31, 2023 at 5:33 am
real cialis on line ordering: generic cialis – canadian meds cialis
August 31, 2023 at 5:47 am
cialis with dapoxetine sale cialis.science cialis 10mg generic
August 31, 2023 at 5:52 am
generic ed drugs: best over the counter ed pills – ed dysfunction treatment
August 31, 2023 at 5:53 am
top erection pills: cheap erectile dysfunction pills – male ed drugs
August 31, 2023 at 5:54 am
best male enhancement pills: best over the counter ed pills – cheap erectile dysfunction pill
August 31, 2023 at 5:55 am
http://cialis.science/# cialis express delivery australia
August 31, 2023 at 6:13 am
Kamagra tablets: Kamagra tablets 100mg – Kamagra Oral Jelly buy online
August 31, 2023 at 6:15 am
best ed treatment pills: ed remedies – new ed treatments
August 31, 2023 at 6:22 am
http://kamagra.men/# kamagra oral jelly
August 31, 2023 at 6:24 am
male ed drugs erection pills online ed pills that really work
August 31, 2023 at 6:33 am
cialis 20mg prices: where can i get cialis overnite – cialis australia express shipping
August 31, 2023 at 6:35 am
non prescription ed drugs: erection pills online – non prescription ed pills
August 31, 2023 at 6:35 am
natural ed medications: best erectile dysfunction pills – ed drugs
August 31, 2023 at 6:48 am
http://kamagra.men/# Kamagra tablets 100mg
August 31, 2023 at 6:55 am
buy kamagra online: Kamagra tablets 100mg – buy kamagra online
August 31, 2023 at 6:56 am
kamagra: cheap kamagra – buy kamagra online
August 31, 2023 at 7:03 am
cialis 20 mg cialis.science cialis without prescriptions uk
August 31, 2023 at 7:14 am
http://kamagra.men/# buy kamagra
August 31, 2023 at 7:16 am
kamagra: order kamagra oral jelly – Kamagra tablets 100mg
August 31, 2023 at 7:20 am
generic cialis without prescription: buy cialis no prescription – generic cialis tadalafil 40 mg
August 31, 2023 at 7:37 am
Kamagra tablets 100mg: Kamagra tablets – buy kamagra online
August 31, 2023 at 7:41 am
http://kamagra.men/# Kamagra Oral Jelly buy online
August 31, 2023 at 7:45 am
otc ed pills male ed pills cheapest ed pills online
August 31, 2023 at 7:57 am
Kamagra tablets 100mg: Kamagra tablets – order kamagra oral jelly
August 31, 2023 at 7:58 am
best medication for ed: erection pills online – erectile dysfunction drugs
August 31, 2023 at 8:05 am
Kamagra tablets 100mg: kamagra oral jelly – Kamagra tablets 100mg
August 31, 2023 at 8:18 am
cialis without a prescription: buy cialis no prescription – cialis without persciction
August 31, 2023 at 8:25 am
buy kamagra online Kamagra Oral Jelly buy online Kamagra tablets 100mg
August 31, 2023 at 8:35 am
http://edpill.men/# best ed pills at gnc
August 31, 2023 at 8:39 am
cialis shop in australia: cialis.science – cialis buy india
August 31, 2023 at 8:40 am
cialis dosage for women: cheap cialis – cialis profesional vs. super active vs. cialis
August 31, 2023 at 8:49 am
cialis pills canada: Buy Cialis online – viagra cialis levitra trial pack
August 31, 2023 at 9:02 am
canadian pharmacy for cialis for sale: cheap cialis – can i buy cialis without prescription pay pal
August 31, 2023 at 9:11 am
canada customs and cialis buy cialis no prescription viagra sampleviagra cialis
August 31, 2023 at 9:15 am
I believe one of your adverts caused my browser to resize, you may well want to put that on your blacklist.
August 31, 2023 at 9:51 am
the best ed pills: ed pills for sale – ed pills that work
August 31, 2023 at 10:09 am
ed drug prices best over the counter ed pills ed treatment review
August 31, 2023 at 10:58 am
kamagra: kamagra – buy kamagra online
August 31, 2023 at 11:07 am
kamagra order kamagra oral jelly cheap kamagra
August 31, 2023 at 12:02 pm
genic cialis: buy cialis no prescription – buy voucher for cialis daily online
August 31, 2023 at 12:06 pm
cialis professional vs cialis cialis.science cialis generic alternative
August 31, 2023 at 1:05 pm
kamagra Kamagra tablets Kamagra tablets
August 31, 2023 at 1:08 pm
cialis onine: buy cialis no prescription – cialis pills for sale
August 31, 2023 at 2:05 pm
pills erectile dysfunction cheap erectile dysfunction pills cheap erectile dysfunction pill
August 31, 2023 at 2:15 pm
buy cialis no prescription: generic cialis – buy cialis 20mg
August 31, 2023 at 3:04 pm
Kamagra tablets buy kamagra buy kamagra
August 31, 2023 at 3:22 pm
Kamagra Oral Jelly buy online: Kamagra tablets – kamagra oral jelly
August 31, 2023 at 10:56 pm
buy generic cialis buy cialis no prescription cialis overnight
August 31, 2023 at 11:08 pm
where to buy cialis online: cialis (generic) – cialis 10mg ireland
August 31, 2023 at 11:49 pm
cialis online reddit generic cialis no prescription paypal buy cheap cialis online with mastercard
September 1, 2023 at 12:13 am
non prescription ed pills: ed pills for sale – cheap ed drugs
September 1, 2023 at 12:46 am
buy kamagra Kamagra tablets Kamagra tablets 100mg
September 1, 2023 at 1:22 am
to buy cialis generic: cheap cialis – cialis no perscription overnight delivery
September 1, 2023 at 1:41 am
order kamagra oral jelly Kamagra tablets Kamagra tablets
September 1, 2023 at 2:22 am
cheap erectile dysfunction pills online: ed pills – drugs for ed
September 1, 2023 at 2:31 am
levitra or cialis which is better cialis.science i want to buy cialis in australia
September 1, 2023 at 3:24 am
cheap erectile dysfunction pills online best over the counter ed pills best ed medication
September 1, 2023 at 3:26 am
cialis price: cialis.science – cialis on line pharm
September 1, 2023 at 4:17 am
buy kamagra online buy kamagra online order kamagra oral jelly
September 1, 2023 at 4:28 am
cheap kamagra: buy kamagra – order kamagra oral jelly
September 1, 2023 at 5:11 am
cialis sell Buy Tadalafil 20mg brand cialis online pharmacy
September 1, 2023 at 6:05 am
buy kamagra order kamagra oral jelly Kamagra tablets 100mg
September 1, 2023 at 6:37 am
best male enhancement pills: ed pills for sale – ed pills cheap
September 1, 2023 at 8:35 am
where can i buy original cialis cheap cialis generic cialis 20mg
September 1, 2023 at 8:50 am
where can i buy cialis 20mg: Buy Cialis online – buy cialis online free shipping
September 1, 2023 at 9:15 am
cialis reviews patients cheap cialis cialis without a script
September 1, 2023 at 9:38 am
Kamagra Oral Jelly buy online: kamagra oral jelly – kamagra
September 1, 2023 at 9:55 am
the best ed pills new treatments for ed best pill for ed
September 1, 2023 at 10:24 am
best ed treatment pills: ed pills non prescription – medicine for erectile
September 1, 2023 at 10:34 am
pills erectile dysfunction cheap erectile dysfunction pills best drug for ed
September 1, 2023 at 11:10 am
cialis with dapoxetine overnite: cialis.science – generic cialis for daily use
September 1, 2023 at 11:13 am
cialis drug Cialis without a doctor prescription cialis without prescriptions canada
September 1, 2023 at 11:52 am
cheap kamagra Kamagra tablets 100mg order kamagra oral jelly
September 1, 2023 at 11:57 am
buy kamagra online: Kamagra Oral Jelly buy online – Kamagra tablets 100mg
September 1, 2023 at 12:32 pm
ed medication online ed pills best medication for ed
September 1, 2023 at 12:45 pm
cialis online mastercard: Buy Tadalafil 20mg – viagra vs cialis side effects
September 1, 2023 at 1:12 pm
kamagra oral jelly Kamagra Oral Jelly buy online kamagra oral jelly
September 1, 2023 at 1:31 pm
where to buy cialis with paypal: cheap cialis with dapoxetine – cialis pills canada
September 1, 2023 at 2:19 pm
cheap kamagra: Kamagra tablets – Kamagra Oral Jelly buy online
September 1, 2023 at 2:33 pm
ed treatment review ed pills medicine erectile dysfunction
September 1, 2023 at 3:48 pm
ed drugs list best over the counter ed pills ed meds online without doctor prescription
September 1, 2023 at 3:50 pm
mens ed pills: erection pills online – non prescription ed pills
September 1, 2023 at 4:19 pm
buy kamagra online Kamagra Oral Jelly buy online buy kamagra
September 1, 2023 at 4:30 pm
cialis dosage recommend: cheap cialis – 36 hour cialis online
September 1, 2023 at 5:01 pm
cialis with dapoxetine online buy cialis no prescription how much is cialis without insurance
September 1, 2023 at 5:19 pm
30 day free trial cialis: cialis.science – buy brand cialis online usa
September 1, 2023 at 5:41 pm
gnc ed pills ed pills non prescription the best ed pill
September 1, 2023 at 6:06 pm
Kamagra tablets 100mg: kamagra – kamagra
September 1, 2023 at 6:22 pm
what are ed drugs best non prescription ed pills best ed drug
September 1, 2023 at 6:52 pm
use paypal to buy cialis generic cialis germany cialis professional
September 1, 2023 at 7:22 pm
cialis delivered in 24 hours cialis.science best cialis price
September 1, 2023 at 8:13 pm
When I originally commented I clicked the “Notify me when new comments are added” checkbox and now each time a comment is added I get three e-mails with the same comment. Is there any way you can remove me from that service? Many thanks!
September 2, 2023 at 5:43 pm
I just added this site to my feed reader, great stuff. Can not get enough!
September 2, 2023 at 8:54 pm
ivermectin new zealand: stromectol ivermectin 3 mg – ivermectin 12 mg
September 2, 2023 at 8:56 pm
https://cytotec.auction/# buy cytotec online fast delivery
September 2, 2023 at 9:07 pm
buy cytotec online: Cytotec 200mcg price – buy cytotec in usa
September 2, 2023 at 9:11 pm
Misoprostol 200 mg buy online: Cytotec 200mcg price – buy misoprostol over the counter
September 2, 2023 at 9:12 pm
stromectol 3 mg price ivermectin usa price stromectol australia
September 2, 2023 at 9:13 pm
order cytotec online: buy cytotec pills online cheap – cytotec abortion pill
September 2, 2023 at 9:16 pm
http://cytotec.auction/# cytotec abortion pill
September 2, 2023 at 9:26 pm
ivermectin pills: ivermectin cost canada – buy stromectol online uk
September 2, 2023 at 9:33 pm
buy cytotec online fast delivery: cytotec online – cytotec abortion pill
September 2, 2023 at 9:38 pm
https://ivermectin.auction/# ivermectin canada
September 2, 2023 at 9:45 pm
order cytotec online: buy cytotec pills online cheap – buy cytotec pills online cheap
September 2, 2023 at 9:48 pm
neurontin 100 mg tablets neurontin cost in canada how much is neurontin
September 2, 2023 at 9:53 pm
neurontin drug: buy neurontin canada – canada neurontin 100mg lowest price
September 2, 2023 at 9:59 pm
https://ivermectin.auction/# ivermectin 3mg tablets price
September 2, 2023 at 10:04 pm
Abortion pills online: buy misoprostol over the counter – cytotec online
September 2, 2023 at 10:12 pm
stromectol buy uk: ivermectin syrup – stromectol south africa
September 2, 2023 at 10:13 pm
ivermectin 6 mg tablets: ivermectin usa – stromectol otc
September 2, 2023 at 10:21 pm
http://gabapentin.tech/# cheap neurontin online
September 2, 2023 at 10:23 pm
gabapentin 600 mg: buy gabapentin online – cost of neurontin 100mg
September 2, 2023 at 10:33 pm
purchase cytotec: Abortion pills online – Cytotec 200mcg price
September 2, 2023 at 10:42 pm
neurontin 800 mg: purchase neurontin – ordering neurontin online
September 2, 2023 at 10:43 pm
http://gabapentin.tech/# neurontin prescription online
September 2, 2023 at 10:44 pm
stromectol medicine: stromectol 3mg tablets – price of ivermectin
September 2, 2023 at 10:52 pm
neurontin for sale: neurontin capsules 100mg – neurontin capsule 600mg
September 2, 2023 at 11:01 pm
neurontin 100 mg tablets neurontin brand name in india neurontin cap
September 2, 2023 at 11:01 pm
ivermectin 90 mg: ivermectin uk – buy ivermectin
September 2, 2023 at 11:06 pm
http://cytotec.auction/# order cytotec online
September 2, 2023 at 11:13 pm
cytotec buy online usa: cytotec buy online usa – buy misoprostol over the counter
September 2, 2023 at 11:14 pm
buy cytotec online: order cytotec online – buy cytotec online
September 2, 2023 at 11:20 pm
cytotec buy online usa: buy misoprostol over the counter – Abortion pills online
September 2, 2023 at 11:27 pm
http://ivermectin.auction/# buy liquid ivermectin
September 2, 2023 at 11:33 pm
gabapentin buy: neurontin 100mg discount – neurontin price in india
September 2, 2023 at 11:37 pm
neurontin 500 mg neurontin cost generic neurontin 300 mg capsule
September 2, 2023 at 11:39 pm
generic neurontin 600 mg: drug neurontin 20 mg – buy gabapentin
September 2, 2023 at 11:44 pm
cytotec pills buy online: buy cytotec over the counter – buy cytotec pills online cheap
September 2, 2023 at 11:47 pm
https://cytotec.auction/# buy cytotec over the counter
September 2, 2023 at 11:52 pm
gabapentin buy: neurontin 100mg cost – buy gabapentin online
September 2, 2023 at 11:58 pm
Abortion pills online: buy cytotec over the counter – buy cytotec online fast delivery
September 3, 2023 at 12:12 am
cost of stromectol medication buy ivermectin canada ivermectin 6 mg tablets
September 3, 2023 at 12:12 am
neurontin 2400 mg: neurontin buy from canada – neurontin from canada
September 3, 2023 at 12:13 am
ivermectin 6mg dosage: stromectol cost – stromectol cvs
September 3, 2023 at 12:18 am
neurontin 100 mg capsule: where can i buy neurontin from canada – neurontin pfizer
September 3, 2023 at 12:25 am
http://cytotec.auction/# buy cytotec in usa
September 3, 2023 at 12:33 am
stromectol 0.5 mg: ivermectin purchase – ivermectin 1 cream
September 3, 2023 at 12:37 am
ivermectin generic cream: stromectol over the counter – ivermectin 250ml
September 3, 2023 at 12:42 am
ivermectin 4: ivermectin pills human – stromectol tab price
September 3, 2023 at 12:45 am
https://ivermectin.auction/# ivermectin 10 ml
September 3, 2023 at 12:46 am
ivermectin 1 cream stromectol australia can you buy stromectol over the counter
September 3, 2023 at 12:53 am
ivermectin over the counter: cost of ivermectin – stromectol otc
September 3, 2023 at 12:56 am
buy misoprostol over the counter: buy cytotec pills online cheap – cytotec buy online usa
September 3, 2023 at 1:04 am
https://cytotec.auction/# buy cytotec online fast delivery
September 3, 2023 at 1:11 am
drug neurontin 20 mg: neurontin 100mg caps – neurontin 4000 mg
September 3, 2023 at 1:13 am
Cytotec 200mcg price: cytotec buy online usa – Cytotec 200mcg price
September 3, 2023 at 1:15 am
ivermectin ebay: stromectol tablets uk – stromectol tablets buy online
September 3, 2023 at 1:20 am
ivermectin pills canada stromectol australia ivermectin buy nz
September 3, 2023 at 1:23 am
http://cytotec.auction/# cytotec online
September 3, 2023 at 1:32 am
neurontin 300 mg pill: neurontin capsules 300mg – canada neurontin 100mg discount
September 3, 2023 at 1:35 am
buy cytotec pills: cytotec abortion pill – buy cytotec over the counter
September 3, 2023 at 1:40 am
ivermectin 250ml: stromectol australia – ivermectin human
September 3, 2023 at 1:42 am
https://ivermectin.auction/# ivermectin 2ml
September 3, 2023 at 1:52 am
Misoprostol 200 mg buy online: purchase cytotec – buy cytotec
September 3, 2023 at 1:54 am
purchase oral ivermectin: ivermectin 50ml – ivermectin new zealand
September 3, 2023 at 2:01 am
http://gabapentin.tech/# buy neurontin online no prescription
September 3, 2023 at 2:10 am
stromectol uk buy: stromectol nz – stromectol australia
September 3, 2023 at 2:12 am
neurontin 800 mg pill: buy neurontin 100 mg – drug neurontin
September 3, 2023 at 2:13 am
stromectol cvs: ivermectin 5 mg price – ivermectin price uk
September 3, 2023 at 2:20 am
https://cytotec.auction/# buy misoprostol over the counter
September 3, 2023 at 2:28 am
cytotec online purchase cytotec cytotec abortion pill
September 3, 2023 at 2:32 am
neurontin 300 mg pill: neurontin 600 mg – neurontin 100mg discount
September 3, 2023 at 2:32 am
ivermectin tablets order: stromectol 15 mg – stromectol buy
September 3, 2023 at 2:39 am
http://cytotec.auction/# cytotec buy online usa
September 3, 2023 at 2:52 am
ivermectin online: ivermectin 3 mg tabs – ivermectin coronavirus
September 3, 2023 at 2:58 am
http://cytotec.auction/# buy cytotec online fast delivery
September 3, 2023 at 3:02 am
ivermectin nz ivermectin stromectol ivermectin lotion
September 3, 2023 at 3:08 am
ivermectin 10 ml: ivermectin cost – buy stromectol canada
September 3, 2023 at 3:11 am
п»їcytotec pills online: cytotec buy online usa – purchase cytotec
September 3, 2023 at 3:12 am
cytotec online: buy misoprostol over the counter – Cytotec 200mcg price
September 3, 2023 at 3:17 am
http://ivermectin.auction/# ivermectin stromectol
September 3, 2023 at 3:30 am
buy cytotec pills: buy cytotec – order cytotec online
September 3, 2023 at 3:32 am
neurontin rx: neurontin tablets uk – drug neurontin
September 3, 2023 at 3:37 am
ivermectin generic cream: stromectol for sale – ivermectin 9 mg
September 3, 2023 at 3:49 am
п»їcytotec pills online: buy cytotec online – Misoprostol 200 mg buy online
September 3, 2023 at 3:52 am
ivermectin 500mg: stromectol 12mg – п»їorder stromectol online
September 3, 2023 at 3:56 am
http://gabapentin.tech/# neurontin 100mg tablet
September 3, 2023 at 4:06 am
neurontin drug: gabapentin 100mg – neurontin 100mg tablet
September 3, 2023 at 4:08 am
stromectol without prescription: where to buy stromectol – ivermectin otc
September 3, 2023 at 4:15 am
http://ivermectin.auction/# stromectol in canada
September 3, 2023 at 4:30 am
buy stromectol canada: stromectol 3 mg – stromectol order
September 3, 2023 at 4:33 am
https://gabapentin.tech/# neurontin coupon
September 3, 2023 at 4:34 am
buy cytotec over the counter: Abortion pills online – buy misoprostol over the counter
September 3, 2023 at 4:44 am
ivermectin pills canada ivermectin 1mg stromectol tablets
September 3, 2023 at 4:50 am
buy cytotec over the counter: п»їcytotec pills online – buy cytotec online
September 3, 2023 at 5:03 am
medication neurontin 300 mg: buy neurontin online – neurontin tablets
September 3, 2023 at 5:05 am
neurontin mexico: neurontin 800 mg cost – neurontin 300 mg price
September 3, 2023 at 5:09 am
stromectol price us: ivermectin humans – stromectol medication
September 3, 2023 at 5:11 am
http://ivermectin.auction/# stromectol ivermectin buy
September 3, 2023 at 5:25 am
neurontin 100 mg cap: neurontin prices generic – neurontin 800 mg price
September 3, 2023 at 5:29 am
Abortion pills online: buy cytotec over the counter – buy cytotec over the counter
September 3, 2023 at 5:31 am
http://gabapentin.tech/# neurontin uk
September 3, 2023 at 5:32 am
buy cytotec pills online cheap: cytotec buy online usa – order cytotec online
September 3, 2023 at 5:44 am
buy cytotec over the counter: buy cytotec pills online cheap – buy cytotec pills
September 3, 2023 at 5:49 am
neurontin cost australia: neurontin price uk – neurontin 800 mg price
September 3, 2023 at 5:53 am
buy gabapentin online neurontin price comparison neurontin medicine
September 3, 2023 at 6:01 am
neurontin 300mg: neurontin 800 mg tablet – neurontin 500 mg
September 3, 2023 at 6:04 am
cytotec online: cytotec pills buy online – buy misoprostol over the counter
September 3, 2023 at 6:09 am
ivermectin online: ivermectin price – stromectol for humans
September 3, 2023 at 6:10 am
http://cytotec.auction/# order cytotec online
September 3, 2023 at 6:23 am
Misoprostol 200 mg buy online: buy cytotec over the counter – order cytotec online
September 3, 2023 at 6:28 am
neurontin 600 mg tablet neurontin price in india neurontin 100mg capsule price
September 3, 2023 at 6:29 am
neurontin 900 mg: can i buy neurontin over the counter – neurontin 100mg cost
September 3, 2023 at 6:31 am
where to buy ivermectin pills: buy stromectol canada – ivermectin buy
September 3, 2023 at 6:43 am
neurontin 100mg cost: ordering neurontin online – neurontin 200 mg
September 3, 2023 at 6:49 am
buy cytotec over the counter: buy cytotec – buy cytotec in usa
September 3, 2023 at 7:00 am
buy gabapentin: neurontin tablets no script – neurontin 400 mg
September 3, 2023 at 7:02 am
buy cheap neurontin online: 2000 mg neurontin – medicine neurontin capsules
September 3, 2023 at 7:03 am
Misoprostol 200 mg buy online buy cytotec online buy cytotec over the counter
September 3, 2023 at 7:08 am
http://ivermectin.auction/# ivermectin humans
September 3, 2023 at 7:14 am
ivermectin humans: ivermectin 10 mg – ivermectin cream 5%
September 3, 2023 at 7:21 am
ivermectin 50ml: stromectol 3mg cost – ivermectin for sale
September 3, 2023 at 7:28 am
http://ivermectin.auction/# ivermectin buy nz
September 3, 2023 at 7:29 am
buy stromectol pills: stromectol 0.5 mg – ivermectin for humans
September 3, 2023 at 7:41 am
neurontin pills for sale: canada neurontin 100mg discount – neurontin 300 mg mexico
September 3, 2023 at 8:22 am
buy ivermectin cream ivermectin 400 mg brands ivermectin 1mg
September 3, 2023 at 9:35 am
order cytotec online buy cytotec online fast delivery п»їcytotec pills online
September 3, 2023 at 10:48 am
buy cytotec in usa buy cytotec online fast delivery buy cytotec pills online cheap
September 3, 2023 at 12:01 pm
cytotec pills buy online order cytotec online purchase cytotec
September 3, 2023 at 2:27 pm
ivermectin nz stromectol prices ivermectin cream uk
September 3, 2023 at 3:42 pm
neurontin online usa neurontin 600 mg capsule neurontin canada
September 3, 2023 at 4:59 pm
generic neurontin pill cheap neurontin online gabapentin online
September 3, 2023 at 8:42 pm
п»їcytotec pills online buy cytotec pills buy misoprostol over the counter
September 3, 2023 at 9:57 pm
cytotec online buy cytotec pills online cheap purchase cytotec
September 3, 2023 at 11:10 pm
prescription medication neurontin generic neurontin pill generic neurontin cost
September 4, 2023 at 12:16 am
buy cytotec in usa Misoprostol 200 mg buy online buy cytotec in usa
September 4, 2023 at 1:24 am
neurontin capsules 600mg neurontin 600 mg cost neurontin 100mg cost
September 4, 2023 at 2:36 am
neurontin 800 mg tablet neurontin pills buy neurontin
September 4, 2023 at 3:52 am
how much is generic neurontin buy neurontin online no prescription neurontin 150mg
September 4, 2023 at 4:49 am
Thanks for your concepts. One thing really noticed is the fact banks plus financial institutions understand the spending routines of consumers and as well understand that the majority of people max away their own credit cards around the holiday seasons. They properly take advantage of that fact and commence flooding ones inbox along with snail-mail box with hundreds of Zero APR credit card offers shortly when the holiday season finishes. Knowing that if you’re like 98 in the American community, you’ll soar at the possible opportunity to consolidate financial debt and switch balances to 0 APR credit cards.
September 4, 2023 at 5:09 am
how much does ivermectin cost ivermectin 1 cream 45gm stromectol 3 mg
September 4, 2023 at 6:22 am
ivermectin uk coronavirus stromectol cream ivermectin 1mg
September 4, 2023 at 7:36 am
oral ivermectin cost stromectol canada ivermectin cream
September 4, 2023 at 8:11 am
Good ? I should certainly pronounce, impressed with your web site. I had no trouble navigating through all tabs and related information ended up being truly simple to do to access. I recently found what I hoped for before you know it at all. Quite unusual. Is likely to appreciate it for those who add forums or anything, website theme . a tones way for your client to communicate. Excellent task..
September 4, 2023 at 8:49 am
neurontin 400 mg cost price of neurontin canada neurontin 100mg discount
September 4, 2023 at 10:02 am
buy generic neurontin neurontin 100 mg tablets neurontin 100 mg tablets
September 4, 2023 at 11:14 am
neurontin medication neurontin 800 neurontin 900
September 4, 2023 at 12:33 pm
neurontin 100 mg neurontin pills neurontin tablets
September 4, 2023 at 1:48 pm
neurontin generic south africa neurontin prescription medication purchase neurontin online
September 4, 2023 at 3:05 pm
cytotec online buy misoprostol over the counter cytotec abortion pill
September 4, 2023 at 4:18 pm
cytotec online cytotec abortion pill cytotec abortion pill
September 4, 2023 at 5:31 pm
neurontin 200 mg tablets neurontin 200 mg capsules neurontin coupon
September 4, 2023 at 6:46 pm
cost of ivermectin cream stromectol 3 mg tablets price stromectol 3 mg tablet
September 4, 2023 at 8:01 pm
over the counter neurontin neurontin capsules 300mg neurontin 202
September 4, 2023 at 9:17 pm
buy neurontin uk neurontin 500 mg tablet generic neurontin cost
September 4, 2023 at 11:47 pm
neurontin price india canada neurontin 100mg discount neurontin canada online
September 5, 2023 at 1:34 am
After research a number of of the weblog posts in your web site now, and I actually like your means of blogging. I bookmarked it to my bookmark website checklist and will likely be checking back soon. Pls try my web page as properly and let me know what you think.
September 5, 2023 at 2:01 am
It?s arduous to seek out knowledgeable people on this matter, however you sound like you recognize what you?re talking about! Thanks
September 5, 2023 at 2:16 am
Cytotec 200mcg price buy cytotec pills Cytotec 200mcg price
September 5, 2023 at 3:30 am
ivermectin syrup generic ivermectin stromectol cvs
September 5, 2023 at 4:43 am
buy cytotec over the counter cytotec pills buy online purchase cytotec
September 5, 2023 at 5:59 am
buy cytotec in usa buy misoprostol over the counter cytotec abortion pill
September 5, 2023 at 7:14 am
brand neurontin 100 mg canada neurontin medicine neurontin 500 mg
September 5, 2023 at 8:28 am
buy cytotec pills online cheap Cytotec 200mcg price order cytotec online
September 5, 2023 at 9:45 am
neurontin cap 300mg price neurontin canada online neurontin cost uk
September 17, 2023 at 7:49 am
Actually, you make a really good webmaster.
The speed with which the website loads is incredible. You almost have the impression of pulling off some unusual trick. Additionally, the contents are masterpieces.
You did a fantastic job researching this subject!
September 17, 2023 at 7:50 am
Actually, you make a really good webmaster.
The speed with which the website loads is incredible. You almost have the impression of pulling off some unusual trick. Additionally, the contents are masterpieces.
You did a fantastic job researching this subject!
September 17, 2023 at 8:03 am
Actually, you make a great webmaster.
The website loads incredibly quickly. You almost have the impression of pulling off some special trick. The contents are a gem, too.
You did a fantastic job on this subject!
September 17, 2023 at 8:07 am
Actually, you do a great job managing your website.
It loads the website incredibly quickly. It almost seems like you’re pulling off some elaborate ruse. The contents are a work of art as well.
This topic has been handled really well by you!
September 17, 2023 at 8:07 am
Actually, you make a really good webmaster.
The speed with which the website loads is incredible. You almost have the impression of pulling off some unusual trick. Additionally, the contents are masterpieces.
You did a fantastic job researching this subject!
September 17, 2023 at 8:09 am
Actually, you make a great webmaster.
The website loads incredibly quickly. You almost have the impression of pulling off some special trick. The contents are a gem, too.
You did a fantastic job on this subject!