Alcohol Dehydrogenase
Alcohol dehydrogenase (ADH) is located in the cytosol of stomach and liver cells and functions as the main enzyme for alcohol metabolism (5). ADH has a low Km and becomes saturated, reaching its Vmax, even at low concentrations of ethanol. Therefore, the enzyme appears to show zero-order kinetics because once the enzyme is saturated, the reaction rate is no longer dictated by the concentration of the ethanol (3).
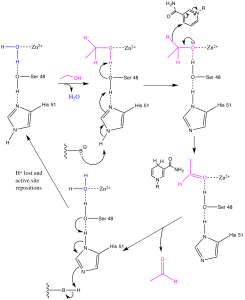
Figure 5: Mechanism of alcohol dehydrogenase. Note that the Zinc atom is coordinated in the active site by Cys-174, Cys-46 and His-67, however, these residues were left out of the mechanism to emphasize the active residues.
ADH is an oxidoreductase enzyme that oxidizes alcohol to acetaldehyde while subsequently reducing an NAD+ cofactor to NADH. A Zn2+ atom is coordinated in the active site by Cys-174, Cys-46 and His-67 and functions to position the alcohol group of ethanol in the active site. Ser-48 and His-51 function similarly to a catalytic dyad, acting as a charge-relay network to help deprotonate the ethanol and activate it to be oxidized to the aldehyde. Before ethanol enters, a water molecule is initially positioned in the active site, but dissociates when the ethanol enters. At the end of the mechanism, water again enters the active site when the oxidized substrate—acetaldehyde—leaves (6).
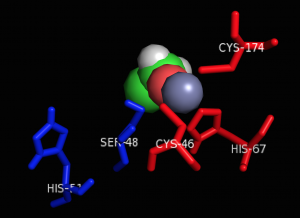
Figure 6: The active site of ADH-1. The zinc atom (purple) coordinates with an ethanol molecule as described above, with His-51 and Ser-48 shown in blue and Cys-174, Cys-46, and His-67 shown in red (4).

Figure 7: Another view of the active site of ADH-1, depicting a side view of ethanol (red/green/white) coordinating with the zinc molecule (purple) (4).
November 10, 2016 at 4:11 pm
Nice figures! This page is nice and concise too, the paragraphs before and after the mechanism figure are well written, and clarify the figures well without adding any confusing or extraneous information.
November 10, 2016 at 5:22 pm
Great page! Particularly liked the part where you compare ADH to a catalytic diad and “charge relay network” – very nicely but also very concisely phrased.
August 1, 2023 at 5:56 am
rcaeeiro http://para-mayores.es/# jtqmfdcx
August 6, 2023 at 6:46 am
accutane australia buy https://isotretinoinacne.shop/# where can i buy accutane from
August 19, 2023 at 11:56 am
tesco pharmacy tadalafil tadalafilise.cyou/#
August 22, 2023 at 3:53 am
Thanks for your posting. I also feel that laptop computers have become more and more popular today, and now tend to be the only form of computer employed in a household. It is because at the same time they are becoming more and more reasonably priced, their processing power keeps growing to the point where they can be as powerful as desktop out of just a few in years past.
August 22, 2023 at 10:09 am
reviews on cialis vs viagra cialis 100 mg usa shopping for cialis
August 23, 2023 at 7:23 pm
It’s a pity you don’t have a donate button! I’d without a doubt donate to this excellent blog! I suppose for now i’ll settle for book-marking and adding your RSS feed to my Google account. I look forward to fresh updates and will talk about this blog with my Facebook group. Chat soon!
August 24, 2023 at 11:30 am
tablete cialis tadalafil insuffisance cardiaque droite cialis commercial
August 25, 2023 at 1:31 am
okmark your blog and check again here regularly. I’m quite certain I will learn many new stuff right here! Good luck for the next!
August 25, 2023 at 3:42 pm
Howdy just wanted to give you a quick heads up and let you know a few of the images aren’t loading correctly. I’m not sure why but I think its a linking issue. I’ve tried it in two different browsers and both show the same results.
August 25, 2023 at 4:16 pm
What i don’t understood is if truth be told how you’re not really much more neatly-favored than you may be right now. You are very intelligent. You already know therefore considerably in terms of this topic, made me personally believe it from numerous various angles. Its like men and women aren’t interested unless it?s something to accomplish with Woman gaga! Your own stuffs nice. All the time handle it up!
August 26, 2023 at 9:31 am
otc cialis usa cialis c20 pill cialis tablete srbija
August 27, 2023 at 2:47 am
Thanks for the suggestions shared using your blog. Something also important I would like to state is that fat reduction is not about going on a celebrity diet and trying to reduce as much weight as you can in a few days. The most effective way to shed weight is by consuming it slowly but surely and following some basic suggestions which can assist you to make the most from your attempt to lose fat. You may realize and already be following most of these tips, yet reinforcing expertise never damages.
August 27, 2023 at 3:24 pm
Thanks for helping me to achieve new thoughts about personal computers. I also hold the belief that certain of the best ways to maintain your notebook computer in excellent condition is to use a hard plastic-type case, or perhaps shell, that fits over the top of your computer. Most of these protective gear tend to be model distinct since they are manufactured to fit perfectly in the natural covering. You can buy all of them directly from the seller, or through third party sources if they are for your notebook, however don’t assume all laptop could have a spend on the market. Just as before, thanks for your suggestions.
August 28, 2023 at 4:58 am
is expired cialis still effective eli lilly tadalafil cialis pharmacy australia
August 28, 2023 at 6:16 am
Howdy just wanted to give you a quick heads up. The words in your article seem to be running off the screen in Safari. I’m not sure if this is a formatting issue or something to do with web browser compatibility but I figured I’d post to let you know. The design look great though! Hope you get the problem solved soon. Many thanks
August 28, 2023 at 3:31 pm
I?m not sure where you’re getting your information, but great topic. I needs to spend some time learning more or understanding more. Thanks for magnificent info I was looking for this information for my mission.
August 29, 2023 at 2:46 am
canada online pharmacy https://certifiedcanadapills.pro/# canadian drug pharmacy
August 29, 2023 at 7:55 am
mexico pharmacies prescription drugs mexican mail order pharmacies mexican mail order pharmacies
August 29, 2023 at 7:57 am
77 canadian pharmacy http://certifiedcanadapills.pro/# canadian pharmacy online
August 29, 2023 at 7:59 am
https://mexicanpharmacy.guru/# mexican mail order pharmacies
August 29, 2023 at 8:10 am
mexican rx online: mexican border pharmacies shipping to usa – purple pharmacy mexico price list
August 29, 2023 at 8:12 am
canadian discount pharmacy prescription drugs canada buy online canada cloud pharmacy
August 29, 2023 at 8:17 am
https://certifiedcanadapills.pro/# canadian drug
August 29, 2023 at 8:34 am
https://certifiedcanadapills.pro/# canada discount pharmacy
August 29, 2023 at 8:40 am
mexican rx online: buying prescription drugs in mexico – mexican mail order pharmacies
August 29, 2023 at 8:48 am
cross border pharmacy canada buy canadian drugs reliable canadian pharmacy
August 29, 2023 at 8:53 am
http://indiapharmacy.cheap/# Online medicine order
August 29, 2023 at 8:58 am
п»їbest mexican online pharmacies: mexican drugstore online – mexican border pharmacies shipping to usa
August 29, 2023 at 9:06 am
safe canadian pharmacy canadian world pharmacy canadian pharmacy 24h com
August 29, 2023 at 9:10 am
https://mexicanpharmacy.guru/# purple pharmacy mexico price list
August 29, 2023 at 9:24 am
pharmacies in mexico that ship to usa buying from online mexican pharmacy medicine in mexico pharmacies
August 29, 2023 at 9:26 am
mexico drug stores pharmacies: mexican pharmaceuticals online – mexican online pharmacies prescription drugs
August 29, 2023 at 9:33 am
top 10 pharmacies in india: reputable indian online pharmacy – canadian pharmacy india
August 29, 2023 at 9:42 am
indianpharmacy com buy prescription drugs from india world pharmacy india
August 29, 2023 at 9:45 am
https://indiapharmacy.cheap/# best india pharmacy
August 29, 2023 at 10:02 am
http://indiapharmacy.cheap/# top 10 pharmacies in india
August 29, 2023 at 10:04 am
top 10 pharmacies in india: best india pharmacy – india pharmacy mail order
August 29, 2023 at 10:08 am
pharmacy wholesalers canada: reputable canadian online pharmacies – online canadian pharmacy
August 29, 2023 at 10:17 am
mexican border pharmacies shipping to usa mexican drugstore online buying from online mexican pharmacy
August 29, 2023 at 10:20 am
https://mexicanpharmacy.guru/# mexico drug stores pharmacies
August 29, 2023 at 10:26 am
indian pharmacy online: india online pharmacy – canadian pharmacy india
August 29, 2023 at 10:29 am
buying drugs from canada https://certifiedcanadapills.pro/# pharmacy com canada
August 29, 2023 at 10:35 am
canadian pharmacy mall canada drugs online review canadian pharmacy review
August 29, 2023 at 10:38 am
http://indiapharmacy.cheap/# Online medicine home delivery
August 29, 2023 at 10:42 am
mexican border pharmacies shipping to usa: mexican mail order pharmacies – п»їbest mexican online pharmacies
August 29, 2023 at 10:44 am
india pharmacy mail order: indian pharmacy – indian pharmacy online
August 29, 2023 at 10:54 am
reputable mexican pharmacies online pharmacies in mexico that ship to usa buying prescription drugs in mexico
August 29, 2023 at 11:02 am
mexican rx online: pharmacies in mexico that ship to usa – mexico drug stores pharmacies
August 29, 2023 at 11:13 am
п»їlegitimate online pharmacies india india online pharmacy top 10 online pharmacy in india
August 29, 2023 at 11:15 am
http://certifiedcanadapills.pro/# my canadian pharmacy
August 29, 2023 at 11:20 am
buying from online mexican pharmacy: mexico drug stores pharmacies – mexican online pharmacies prescription drugs
August 29, 2023 at 11:20 am
mexico drug stores pharmacies: medication from mexico pharmacy – best online pharmacies in mexico
August 29, 2023 at 11:32 am
vipps canadian pharmacy canadian pharmacy canada drugs
August 29, 2023 at 11:33 am
https://mexicanpharmacy.guru/# mexico drug stores pharmacies
August 29, 2023 at 11:38 am
best online pharmacy india: reputable indian online pharmacy – best online pharmacy india
August 29, 2023 at 11:51 am
canadian online drugstore canadian pharmacy 1 internet online drugstore my canadian pharmacy
August 29, 2023 at 11:51 am
https://indiapharmacy.cheap/# top online pharmacy india
August 29, 2023 at 11:56 am
indianpharmacy com: india online pharmacy – indian pharmacy online
August 29, 2023 at 11:58 am
medicine in mexico pharmacies: reputable mexican pharmacies online – mexico drug stores pharmacies
August 29, 2023 at 12:10 pm
buy medicines online in india indian pharmacy paypal india pharmacy mail order
August 29, 2023 at 12:14 pm
buying prescription drugs in mexico online: buying prescription drugs in mexico – mexico drug stores pharmacies
August 29, 2023 at 12:27 pm
http://certifiedcanadapills.pro/# canadian pharmacy store
August 29, 2023 at 12:28 pm
canadian family pharmacy canada ed drugs cheap canadian pharmacy
August 29, 2023 at 12:31 pm
pharmacies in mexico that ship to usa: mexican border pharmacies shipping to usa – mexican drugstore online
August 29, 2023 at 12:36 pm
Online medicine home delivery: world pharmacy india – reputable indian online pharmacy
August 29, 2023 at 12:44 pm
https://certifiedcanadapills.pro/# best canadian pharmacy online
August 29, 2023 at 12:45 pm
canadianpharmacymeds com canadian drugs legal to buy prescription drugs from canada
August 29, 2023 at 1:02 pm
https://mexicanpharmacy.guru/# mexican online pharmacies prescription drugs
August 29, 2023 at 1:02 pm
canadian pharmacy meds review prescription drugs canada buy online canadian pharmacy king
August 29, 2023 at 1:06 pm
pharmacies in mexico that ship to usa: mexico drug stores pharmacies – mexico drug stores pharmacies
August 29, 2023 at 1:15 pm
mexican online pharmacies prescription drugs: best online pharmacies in mexico – mexico pharmacies prescription drugs
August 29, 2023 at 1:19 pm
https://mexicanpharmacy.guru/# buying prescription drugs in mexico
August 29, 2023 at 1:20 pm
india pharmacy top 10 online pharmacy in india world pharmacy india
August 29, 2023 at 1:25 pm
medicine in mexico pharmacies: mexican rx online – buying prescription drugs in mexico online
August 29, 2023 at 1:38 pm
https://mexicanpharmacy.guru/# mexican online pharmacies prescription drugs
August 29, 2023 at 1:40 pm
Online medicine home delivery best online pharmacy india top 10 online pharmacy in india
August 29, 2023 at 1:45 pm
top 10 pharmacies in india: india pharmacy – indian pharmacy
August 29, 2023 at 1:57 pm
http://mexicanpharmacy.guru/# mexican drugstore online
August 29, 2023 at 1:59 pm
best india pharmacy pharmacy website india cheapest online pharmacy india
August 29, 2023 at 2:04 pm
canadian medications: best canadian online pharmacy – best online canadian pharmacy
August 29, 2023 at 2:19 pm
mexico drug stores pharmacies mexico drug stores pharmacies medication from mexico pharmacy
August 29, 2023 at 2:23 pm
best online pharmacies in mexico: mexico drug stores pharmacies – purple pharmacy mexico price list
August 29, 2023 at 2:34 pm
cheapest online pharmacy india: top 10 pharmacies in india – cheapest online pharmacy india
August 29, 2023 at 2:35 pm
https://indiapharmacy.cheap/# indian pharmacies safe
August 29, 2023 at 2:38 pm
onlinecanadianpharmacy 24 canadian online pharmacy reviews best canadian online pharmacy
August 29, 2023 at 2:42 pm
buy prescription drugs from canada cheap: canadian pharmacy price checker – best canadian pharmacy to buy from
August 29, 2023 at 2:54 pm
https://mexicanpharmacy.guru/# mexican mail order pharmacies
August 29, 2023 at 2:58 pm
india pharmacy mail order best india pharmacy india online pharmacy
August 29, 2023 at 3:02 pm
canada drugstore pharmacy rx: safe reliable canadian pharmacy – canadian 24 hour pharmacy
August 29, 2023 at 3:13 pm
http://indiapharmacy.cheap/# buy prescription drugs from india
August 29, 2023 at 3:13 pm
canadian pharmacy world: canadian pharmacy near me – canadian pharmacy uk delivery
August 29, 2023 at 3:18 pm
canadian pharmacy ratings canadian compounding pharmacy canadian drugs pharmacy
August 29, 2023 at 3:21 pm
medicine in mexico pharmacies: medicine in mexico pharmacies – mexican drugstore online
August 29, 2023 at 3:32 pm
http://certifiedcanadapills.pro/# trustworthy canadian pharmacy
August 29, 2023 at 3:38 pm
mexico drug stores pharmacies buying prescription drugs in mexico buying prescription drugs in mexico
August 29, 2023 at 3:40 pm
thecanadianpharmacy: online pharmacy canada – maple leaf pharmacy in canada
August 29, 2023 at 3:52 pm
https://certifiedcanadapills.pro/# onlinepharmaciescanada com
August 29, 2023 at 3:53 pm
best online pharmacy india: cheapest online pharmacy india – top 10 pharmacies in india
August 29, 2023 at 3:59 pm
vipps canadian pharmacy: best canadian pharmacy to order from – legit canadian pharmacy online
August 29, 2023 at 4:11 pm
http://indiapharmacy.cheap/# pharmacy website india
August 29, 2023 at 4:18 pm
cheapest online pharmacy india buy prescription drugs from india mail order pharmacy india
August 29, 2023 at 4:19 pm
mexican rx online: mexico pharmacies prescription drugs – pharmacies in mexico that ship to usa
August 29, 2023 at 4:31 pm
http://mexicanpharmacy.guru/# mexican online pharmacies prescription drugs
August 29, 2023 at 4:33 pm
reputable indian pharmacies: indian pharmacies safe – online pharmacy india
August 29, 2023 at 4:38 pm
india online pharmacy reputable indian pharmacies top 10 pharmacies in india
August 29, 2023 at 4:52 pm
http://mexicanpharmacy.guru/# purple pharmacy mexico price list
August 29, 2023 at 4:59 pm
mexican rx online mexican online pharmacies prescription drugs mexico pharmacies prescription drugs
August 29, 2023 at 5:01 pm
ed meds online canada: canada discount pharmacy – pharmacy rx world canada
August 29, 2023 at 5:12 pm
https://mexicanpharmacy.guru/# mexican online pharmacies prescription drugs
August 29, 2023 at 5:13 pm
india pharmacy mail order: online shopping pharmacy india – india pharmacy mail order
August 29, 2023 at 5:19 pm
canadian pharmacy canada pharmacy reviews reliable canadian pharmacy
August 29, 2023 at 5:38 pm
reputable mexican pharmacies online medication from mexico pharmacy mexico drug stores pharmacies
August 29, 2023 at 5:40 pm
indian pharmacy online: top 10 pharmacies in india – world pharmacy india
August 29, 2023 at 5:58 pm
medicine in mexico pharmacies buying prescription drugs in mexico online buying prescription drugs in mexico online
August 29, 2023 at 5:59 pm
online pharmacy india: indian pharmacy online – india pharmacy mail order
August 29, 2023 at 6:09 pm
https://indiapharmacy.cheap/# reputable indian pharmacies
August 29, 2023 at 6:18 pm
best online pharmacy india: indian pharmacies safe – mail order pharmacy india
August 29, 2023 at 6:28 pm
https://mexicanpharmacy.guru/# mexico pharmacies prescription drugs
August 29, 2023 at 6:31 pm
is canadian pharmacy legit: canadian world pharmacy – canadian pharmacy sarasota
August 29, 2023 at 6:37 pm
india pharmacy: pharmacy website india – top online pharmacy india
August 29, 2023 at 6:57 pm
п»їbest mexican online pharmacies: best online pharmacies in mexico – purple pharmacy mexico price list
August 29, 2023 at 6:58 pm
mexican border pharmacies shipping to usa п»їbest mexican online pharmacies best online pharmacies in mexico
August 29, 2023 at 7:07 pm
https://certifiedcanadapills.pro/# canada pharmacy
August 29, 2023 at 7:11 pm
pet meds without vet prescription canada: canadian pharmacy uk delivery – legitimate canadian pharmacies
August 29, 2023 at 7:16 pm
best online pharmacy india: best india pharmacy – top online pharmacy india
August 29, 2023 at 7:18 pm
mexico drug stores pharmacies medicine in mexico pharmacies mexican pharmaceuticals online
August 29, 2023 at 7:26 pm
http://certifiedcanadapills.pro/# my canadian pharmacy review
August 29, 2023 at 7:35 pm
indian pharmacy online: indianpharmacy com – reputable indian online pharmacy
August 29, 2023 at 7:45 pm
https://certifiedcanadapills.pro/# buy prescription drugs from canada cheap
August 29, 2023 at 7:50 pm
top 10 online pharmacy in india: reputable indian pharmacies – online pharmacy india
August 29, 2023 at 7:55 pm
buying from online mexican pharmacy: buying from online mexican pharmacy – purple pharmacy mexico price list
August 29, 2023 at 7:59 pm
п»їbest mexican online pharmacies medication from mexico pharmacy buying from online mexican pharmacy
August 29, 2023 at 8:06 pm
http://indiapharmacy.cheap/# buy prescription drugs from india
August 29, 2023 at 8:14 pm
indianpharmacy com: indian pharmacies safe – online shopping pharmacy india
August 29, 2023 at 8:26 pm
http://mexicanpharmacy.guru/# mexico drug stores pharmacies
August 29, 2023 at 8:33 pm
canadian pharmacy 24 com: canadian valley pharmacy – recommended canadian pharmacies
August 29, 2023 at 8:54 pm
indian pharmacies safe: buy medicines online in india – cheapest online pharmacy india
August 30, 2023 at 7:40 am
You made some decent factors there. I regarded on the web for the difficulty and located most people will associate with together with your website.
August 30, 2023 at 10:49 am
exploring danube delta
August 31, 2023 at 4:04 am
purchase brand cialis: cialis daily online – what works better cialis or viagra?
August 31, 2023 at 4:05 am
ed medication online: ed pills – ed pills comparison
August 31, 2023 at 4:05 am
cheap viagra and cialis in australia cheap cialis generic cialis available in usa
August 31, 2023 at 4:11 am
cialis generic reviews: cialis.science – cialis 20mg overnight
August 31, 2023 at 4:11 am
http://cialis.science/# order cialis no prescription
August 31, 2023 at 4:24 am
original cialis pills: Cialis without a doctor prescription – how much is cialis in canada
August 31, 2023 at 4:25 am
order kamagra oral jelly: Kamagra tablets – buy kamagra online
August 31, 2023 at 4:39 am
http://cialis.science/# cialis soft tabs overnight
August 31, 2023 at 4:45 am
erection pills online ed pills for sale medication for ed
August 31, 2023 at 4:45 am
kamagra: kamagra – buy kamagra
August 31, 2023 at 4:57 am
best deals on cialis super active plus online: cheap cialis – cialis 20mg tablets price
August 31, 2023 at 5:03 am
https://cialis.science/# cialis online free shipping
August 31, 2023 at 5:06 am
kamagra oral jelly: kamagra – cheap kamagra
August 31, 2023 at 5:27 am
kamagra oral jelly: cheap kamagra – buy kamagra online
August 31, 2023 at 5:30 am
https://cialis.science/# cialis 20 mg best price
August 31, 2023 at 5:30 am
kamagra oral jelly buy kamagra online Kamagra tablets 100mg
August 31, 2023 at 5:46 am
natural ed medications: ed pills – men’s ed pills
August 31, 2023 at 5:48 am
best ed medication: erection pills online – the best ed pill
August 31, 2023 at 5:58 am
https://kamagra.men/# kamagra
August 31, 2023 at 6:09 am
cialis in melbourne australia: buy cialis no prescription – can you buy cialis without a prescription
August 31, 2023 at 6:09 am
ed pills gnc ed treatment review cheapest ed pills online
August 31, 2023 at 6:26 am
http://kamagra.men/# buy kamagra online
August 31, 2023 at 6:27 am
medicine for impotence: ed pills for sale – best ed medications
August 31, 2023 at 6:29 am
generic ed drugs: erection pills online – best male enhancement pills
August 31, 2023 at 6:47 am
online cialis australia cialis.science generic cialis online
August 31, 2023 at 6:50 am
buy kamagra online: kamagra oral jelly – Kamagra tablets
August 31, 2023 at 6:51 am
https://edpill.men/# ed medication online
August 31, 2023 at 7:10 am
buy shop cialis 20mg: generic cialis – cialis online canada ripoff
August 31, 2023 at 7:11 am
treatments for ed: best over the counter ed pills – top rated ed pills
August 31, 2023 at 7:12 am
order kamagra oral jelly: kamagra – order kamagra oral jelly
August 31, 2023 at 7:17 am
http://edpill.men/# non prescription ed pills
August 31, 2023 at 7:30 am
cialis for daily use generic cialis fed ex overnight delivery cialis
August 31, 2023 at 7:31 am
Kamagra tablets: kamagra oral jelly – buy kamagra online
August 31, 2023 at 7:45 am
http://cialis.science/# cialis canada no prescription
August 31, 2023 at 7:51 am
medications for ed: ed pills non prescription – ed pills
August 31, 2023 at 7:54 am
cialis recreational use: cheap cialis – how to cut 20mg. cialis
August 31, 2023 at 7:58 am
buy kamagra: cheap kamagra – buy kamagra online
August 31, 2023 at 8:09 am
order kamagra oral jelly Kamagra Oral Jelly buy online Kamagra tablets
August 31, 2023 at 8:12 am
http://kamagra.men/# cheap kamagra
August 31, 2023 at 8:15 am
cheap kamagra: buy kamagra – kamagra oral jelly
August 31, 2023 at 8:33 am
cheap kamagra: Kamagra tablets 100mg – Kamagra tablets 100mg
August 31, 2023 at 8:38 am
https://edpill.men/# п»їerectile dysfunction medication
August 31, 2023 at 8:42 am
medicine erectile dysfunction: best over the counter ed pills – ed drugs compared
August 31, 2023 at 8:50 am
cialis online canada Buy Cialis online resaonably priced cialis
August 31, 2023 at 8:54 am
cheap erectile dysfunction: п»їerectile dysfunction medication – generic ed pills
August 31, 2023 at 9:38 am
treatments for ed: cheap erectile dysfunction pills – ed drugs compared
August 31, 2023 at 9:45 am
generic cialis uk online generic cialis viagra cialis comparison
August 31, 2023 at 10:44 am
cialis blac Cialis without a doctor prescription cialis online overnight delivery
August 31, 2023 at 10:45 am
ed drugs: ed pills non prescription – pills erectile dysfunction
August 31, 2023 at 11:51 am
generic ed pills: ed pills – п»їerectile dysfunction medication
August 31, 2023 at 12:42 pm
cheapest ed pills online best over the counter ed pills best pills for ed
August 31, 2023 at 12:57 pm
kamagra oral jelly: buy kamagra online – Kamagra Oral Jelly buy online
August 31, 2023 at 1:42 pm
cheap kamagra Kamagra tablets Kamagra tablets
August 31, 2023 at 2:03 pm
cheap kamagra: Kamagra tablets – kamagra oral jelly
August 31, 2023 at 2:41 pm
order kamagra oral jelly Kamagra tablets 100mg kamagra oral jelly
August 31, 2023 at 3:10 pm
ed meds online without doctor prescription: cheap erectile dysfunction pills – ed medications list
August 31, 2023 at 3:40 pm
cialis women video Cialis without a doctor prescription buy low dose cialis online
August 31, 2023 at 10:34 pm
buy kamagra online Kamagra Oral Jelly buy online kamagra
August 31, 2023 at 10:57 pm
buy kamagra online: buy kamagra – Kamagra Oral Jelly buy online
August 31, 2023 at 11:28 pm
ed pills best over the counter ed pills medications for ed
September 1, 2023 at 12:01 am
purchase cialis in canada: Cialis without a doctor prescription – cialis 20mg uk
September 1, 2023 at 12:23 am
cialis online american express cheap cialis canada pay cialis with pay pal
September 1, 2023 at 1:10 am
Kamagra Oral Jelly buy online: order kamagra oral jelly – buy kamagra online
September 1, 2023 at 1:21 am
kamagra buy kamagra online buy kamagra online
September 1, 2023 at 2:11 am
ed medication ed pills best erectile dysfunction pills
September 1, 2023 at 3:03 am
compare ed drugs ed pills non prescription erection pills that work
September 1, 2023 at 3:06 am
Thanks for your intriguing article. One other problem is that mesothelioma is generally brought on by the breathing of dust from mesothelioma, which is a carcinogenic material. It’s commonly found among workers in the construction industry who may have long exposure to asbestos. It can be caused by moving into asbestos protected buildings for a long time of time, Genes plays an important role, and some people are more vulnerable to the risk when compared with others.
September 1, 2023 at 3:14 am
Kamagra tablets 100mg: buy kamagra online – Kamagra tablets
September 1, 2023 at 3:57 am
order kamagra oral jelly order kamagra oral jelly buy kamagra online
September 1, 2023 at 4:17 am
buy kamagra online: Kamagra Oral Jelly buy online – order kamagra oral jelly
September 1, 2023 at 4:51 am
cialis free trial canada generic cialis which one is better viagra cialis or laverta
September 1, 2023 at 5:22 am
ed meds: ed pills – otc ed pills
September 1, 2023 at 5:45 am
cheap kamagra Kamagra Oral Jelly buy online kamagra
September 1, 2023 at 6:26 am
best otc ed pills: cheap erectile dysfunction pills – best ed medications
September 1, 2023 at 6:38 am
original cialis uk how to buy generic cialis online cialis soft tabs overnight
September 1, 2023 at 8:19 am
canadian pharmacy non rx cialis cialis 10mg ireland cialis where to buy in las vegas nv
September 1, 2023 at 8:42 am
kamagra oral jelly: Kamagra tablets 100mg – order kamagra oral jelly
September 1, 2023 at 8:59 am
how much is cialis in canada cheap cialis cialis generic cambodia
September 1, 2023 at 9:30 am
online ed medications: ed pills – erection pills that work
September 1, 2023 at 9:40 am
ed meds online without doctor prescription ed pills men’s ed pills
September 1, 2023 at 10:15 am
where to buy generic cialis?: cialis.science – where can i buy cialis in canada
September 1, 2023 at 10:18 am
Kamagra Oral Jelly buy online kamagra kamagra
September 1, 2023 at 10:57 am
generic cialis with dapoxetine 80mg x 10 tabs Cialis without a doctor prescription buy cialis cheap prices fast delivery
September 1, 2023 at 11:01 am
cialis manufactured in canada: Buy Cialis online – cialis from canadian pharmacy registerd
September 1, 2023 at 11:36 am
cheap ed pills best over the counter ed pills male erection pills
September 1, 2023 at 11:49 am
cialis lilly rezeptfrei: cheap cialis – buy cialis 36 hour
September 1, 2023 at 12:16 pm
online ed medications ed pills for sale best drug for ed
September 1, 2023 at 12:36 pm
best price cialis supper active: Buy Cialis online – buy brand cialis
September 1, 2023 at 12:56 pm
medicine erectile dysfunction ed pills non prescription what is the best ed pill
September 1, 2023 at 1:24 pm
non prescription ed pills: best over the counter ed pills – best ed pills at gnc
September 1, 2023 at 1:35 pm
ed pills gnc ed pills cure ed
September 1, 2023 at 3:43 pm
cialis mit dapoxetine: Cialis without a doctor prescription – cialis next day delivery
September 1, 2023 at 4:07 pm
buy kamagra online Kamagra tablets Kamagra Oral Jelly buy online
September 1, 2023 at 4:20 pm
top ed drugs: ed pills for sale – ed meds
September 1, 2023 at 4:45 pm
best ed pill erection pills online natural ed remedies
September 1, 2023 at 5:10 pm
free trial cialis: Cialis without a doctor prescription – cialis daily price
September 1, 2023 at 5:25 pm
order kamagra oral jelly Kamagra Oral Jelly buy online Kamagra tablets
September 1, 2023 at 5:58 pm
cialis daily use side effects: Cialis without a doctor prescription – cialis shop
September 1, 2023 at 6:06 pm
cialis pills overnight Buy Tadalafil 20mg cialis with dapoxetine overnight delivery australia
September 1, 2023 at 6:41 pm
is generic cialis from india safe buy cialis no prescription where to buy cialis online
September 1, 2023 at 7:10 pm
canada cialis with dapoxetine cialis.science cialis buy
September 2, 2023 at 8:51 pm
neurontin 300 mg cost: buy neurontin 300 mg – buying neurontin without a prescription
September 2, 2023 at 8:58 pm
http://gabapentin.tech/# buy neurontin
September 2, 2023 at 8:59 pm
cytotec abortion pill: buy cytotec – cytotec abortion pill
September 2, 2023 at 9:02 pm
neurontin canada online: neurontin coupon – drug neurontin 200 mg
September 2, 2023 at 9:05 pm
ivermectin tablets order ivermectin 3 mg tabs stromectol pill
September 2, 2023 at 9:19 pm
http://cytotec.auction/# buy misoprostol over the counter
September 2, 2023 at 9:21 pm
neurontin 100: neurontin 800 mg pill – how much is neurontin pills
September 2, 2023 at 9:29 pm
п»їcytotec pills online: buy cytotec – cytotec online
September 2, 2023 at 9:39 pm
cytotec pills buy online: buy cytotec online – cytotec pills buy online
September 2, 2023 at 9:42 pm
order cytotec online buy cytotec pills buy cytotec online
September 2, 2023 at 9:58 pm
buy cytotec: п»їcytotec pills online – cytotec abortion pill
September 2, 2023 at 10:01 pm
neurontin canada online: neurontin 500 mg – neurontin 300 mg tablets
September 2, 2023 at 10:02 pm
http://cytotec.auction/# cytotec abortion pill
September 2, 2023 at 10:09 pm
buy neurontin online: generic gabapentin – ordering neurontin online
September 2, 2023 at 10:17 pm
cytotec abortion pill: buy cytotec over the counter – Cytotec 200mcg price
September 2, 2023 at 10:18 pm
stromectol ivermectin 3 mg stromectol drug ivermectin usa
September 2, 2023 at 10:24 pm
https://cytotec.auction/# purchase cytotec
September 2, 2023 at 10:32 pm
buy cytotec over the counter: buy misoprostol over the counter – cytotec buy online usa
September 2, 2023 at 10:36 pm
cytotec buy online usa: Abortion pills online – purchase cytotec
September 2, 2023 at 10:46 pm
http://cytotec.auction/# order cytotec online
September 2, 2023 at 10:49 pm
ivermectin iv: ivermectin 4 – buy ivermectin pills
September 2, 2023 at 10:54 pm
where to buy neurontin gabapentin 300 cost of neurontin 600mg
September 2, 2023 at 10:56 pm
800mg neurontin: buying neurontin online – neurontin 1800 mg
September 2, 2023 at 11:03 pm
buy cytotec online: buy cytotec online – buy misoprostol over the counter
September 2, 2023 at 11:08 pm
https://gabapentin.tech/# neurontin capsules 600mg
September 2, 2023 at 11:09 pm
neurontin cost in singapore: neurontin singapore – neurontin 600
September 2, 2023 at 11:15 pm
gabapentin online: gabapentin – discount neurontin
September 2, 2023 at 11:29 pm
п»їorder stromectol online: stromectol liquid – ivermectin eye drops
September 2, 2023 at 11:30 pm
https://ivermectin.auction/# ivermectin 5
September 2, 2023 at 11:31 pm
neurontin generic cost neurontin 100mg capsule price neurontin price uk
September 2, 2023 at 11:34 pm
neurontin 800 mg capsules: neurontin 300 – neurontin 100
September 2, 2023 at 11:49 pm
http://ivermectin.auction/# stromectol medicine
September 2, 2023 at 11:53 pm
buy cytotec in usa: buy cytotec over the counter – buy cytotec online fast delivery
September 3, 2023 at 12:02 am
Abortion pills online: purchase cytotec – buy cytotec online
September 3, 2023 at 12:05 am
Abortion pills online cytotec online buy cytotec pills
September 3, 2023 at 12:08 am
https://cytotec.auction/# buy cytotec in usa
September 3, 2023 at 12:09 am
ivermectin 50 mg: ivermectin 50 mg – stromectol online canada
September 3, 2023 at 12:27 am
https://ivermectin.auction/# ivermectin generic name
September 3, 2023 at 12:29 am
buy cytotec over the counter: cytotec online – cytotec abortion pill
September 3, 2023 at 12:31 am
neurontin pill: buy neurontin online no prescription – buy neurontin canada
September 3, 2023 at 12:31 am
generic gabapentin: neurontin cap 300mg – neurontin 400 mg cost
September 3, 2023 at 12:47 am
https://cytotec.auction/# buy cytotec in usa
September 3, 2023 at 12:49 am
stromectol drug: ivermectin tablets order – cost of ivermectin lotion
September 3, 2023 at 12:51 am
neurontin online usa: gabapentin online – gabapentin 100mg
September 3, 2023 at 1:00 am
neurontin cost in canada: neurontin 800 mg tablets – buy gabapentin online
September 3, 2023 at 1:06 am
http://ivermectin.auction/# ivermectin tablets uk
September 3, 2023 at 1:09 am
buy stromectol online uk: ivermectin 1 – ivermectin 400 mg brands
September 3, 2023 at 1:14 am
neurontin price australia neurontin 600 mg coupon neurontin 150mg
September 3, 2023 at 1:25 am
http://ivermectin.auction/# stromectol 3 mg tablet
September 3, 2023 at 1:28 am
neurontin 100 mg: buying neurontin without a prescription – neurontin capsules 600mg
September 3, 2023 at 1:44 am
http://cytotec.auction/# п»їcytotec pills online
September 3, 2023 at 1:48 am
over the counter neurontin neurontin uk neurontin 300mg capsule
September 3, 2023 at 1:48 am
stromectol generic name: ivermectin price uk – ivermectin drug
September 3, 2023 at 1:58 am
buy cytotec over the counter: п»їcytotec pills online – buy cytotec
September 3, 2023 at 2:08 am
generic gabapentin: neurontin brand name 800 mg – neurontin price
September 3, 2023 at 2:08 am
Cytotec 200mcg price: buy cytotec – cytotec buy online usa
September 3, 2023 at 2:22 am
http://ivermectin.auction/# stromectol oral
September 3, 2023 at 2:27 am
Misoprostol 200 mg buy online: Cytotec 200mcg price – cytotec online
September 3, 2023 at 2:28 am
stromectol order online: ivermectin 4000 mcg – ivermectin 1% cream generic
September 3, 2023 at 2:41 am
https://gabapentin.tech/# neurontin gel
September 3, 2023 at 2:46 am
buy cytotec online fast delivery: buy cytotec pills – cytotec online
September 3, 2023 at 2:48 am
buy cytotec: purchase cytotec – Misoprostol 200 mg buy online
September 3, 2023 at 2:56 am
п»їcytotec pills online buy cytotec over the counter п»їcytotec pills online
September 3, 2023 at 2:57 am
neurontin cost australia: neurontin sale – neurontin 300 mg caps
September 3, 2023 at 3:00 am
https://cytotec.auction/# buy cytotec over the counter
September 3, 2023 at 3:05 am
cytotec online: buy cytotec online – buy cytotec online
September 3, 2023 at 3:08 am
purchase neurontin online: neurontin prices – brand name neurontin
September 3, 2023 at 3:20 am
https://gabapentin.tech/# neurontin buy online
September 3, 2023 at 3:25 am
ivermectin ebay: generic ivermectin cream – stromectol for sale
September 3, 2023 at 3:25 am
cytotec pills buy online: buy cytotec in usa – cytotec online
September 3, 2023 at 3:28 am
stromectol cost: ivermectin cost uk – stromectol ivermectin 3 mg
September 3, 2023 at 3:31 am
buy cytotec pills buy cytotec online fast delivery cytotec abortion pill
September 3, 2023 at 3:39 am
http://cytotec.auction/# buy misoprostol over the counter
September 3, 2023 at 3:44 am
how to get neurontin cheap: buy cheap neurontin online – neurontin
September 3, 2023 at 3:55 am
neurontin 800: neurontin 400 mg price – neurontin 300 mg coupon
September 3, 2023 at 3:58 am
https://cytotec.auction/# buy cytotec
September 3, 2023 at 4:08 am
neurontin 3: buy neurontin uk – neurontin cost in singapore
September 3, 2023 at 4:17 am
http://cytotec.auction/# order cytotec online
September 3, 2023 at 4:23 am
buy generic neurontin: purchase neurontin canada – neurontin 800
September 3, 2023 at 4:27 am
ivermectin 1% cream generic: buy ivermectin nz – ivermectin buy nz
September 3, 2023 at 4:38 am
ivermectin lotion cost stromectol 3mg cost ivermectin oral
September 3, 2023 at 4:41 am
generic gabapentin: neurontin brand name 800 mg – buy neurontin 100 mg
September 3, 2023 at 4:46 am
buy cytotec over the counter: cytotec buy online usa – cytotec online
September 3, 2023 at 4:52 am
neurontin 300 600 mg: price of neurontin – buy neurontin
September 3, 2023 at 4:54 am
http://ivermectin.auction/# ivermectin pills human
September 3, 2023 at 5:00 am
neurontin 600 mg pill: medicine neurontin 300 mg – neurontin tablets no script
September 3, 2023 at 5:06 am
canada where to buy neurontin: neurontin price south africa – neurontin 300 mg mexico
September 3, 2023 at 5:12 am
neurontin 10 mg neurontin 800 mg tablet how much is neurontin
September 3, 2023 at 5:14 am
http://cytotec.auction/# buy cytotec pills
September 3, 2023 at 5:19 am
where to buy ivermectin: ivermectin 3 – stromectol 3 mg
September 3, 2023 at 5:21 am
ivermectin buy australia: ivermectin gel – stromectol prices
September 3, 2023 at 5:25 am
purchase neurontin: buy generic neurontin online – purchase neurontin
September 3, 2023 at 5:33 am
https://ivermectin.auction/# stromectol oral
September 3, 2023 at 5:39 am
order cytotec online: purchase cytotec – п»їcytotec pills online
September 3, 2023 at 5:45 am
buy cytotec over the counter: buy cytotec over the counter – purchase cytotec
September 3, 2023 at 5:47 am
neurontin 900 generic neurontin 600 mg neurontin cost generic
September 3, 2023 at 5:50 am
buy cytotec: Misoprostol 200 mg buy online – п»їcytotec pills online
September 3, 2023 at 5:53 am
https://cytotec.auction/# buy cytotec pills online cheap
September 3, 2023 at 5:58 am
cytotec online: buy misoprostol over the counter – buy cytotec
September 3, 2023 at 6:06 am
buy cytotec over the counter: buy cytotec pills online cheap – п»їcytotec pills online
September 3, 2023 at 6:18 am
neurontin 800 mg pill: prescription drug neurontin – how much is neurontin
September 3, 2023 at 6:19 am
stromectol ivermectin: stromectol online – generic ivermectin
September 3, 2023 at 6:22 am
buy cytotec buy cytotec in usa п»їcytotec pills online
September 3, 2023 at 6:26 am
Misoprostol 200 mg buy online: buy cytotec pills online cheap – buy cytotec over the counter
September 3, 2023 at 6:32 am
https://gabapentin.tech/# neurontin 600 mg cost
September 3, 2023 at 6:37 am
neurontin 500 mg: neurontin 800 mg tablet – neurontin for sale online
September 3, 2023 at 6:46 am
buy cytotec over the counter: buy cytotec online – Misoprostol 200 mg buy online
September 3, 2023 at 6:49 am
neurontin 2018: neurontin canada online – neurontin 900 mg
September 3, 2023 at 6:51 am
http://gabapentin.tech/# neurontin price australia
September 3, 2023 at 6:57 am
cytotec online: cytotec online – order cytotec online
September 3, 2023 at 7:10 am
http://ivermectin.auction/# ivermectin 6 mg tablets
September 3, 2023 at 7:16 am
neurontin 600 mg: neurontin cap – neurontin price
September 3, 2023 at 7:18 am
Misoprostol 200 mg buy online: buy cytotec online – order cytotec online
September 3, 2023 at 7:30 am
https://ivermectin.auction/# generic ivermectin cream
September 3, 2023 at 7:32 am
cytotec abortion pill cytotec online buy misoprostol over the counter
September 3, 2023 at 7:35 am
п»їcytotec pills online: buy cytotec pills online cheap – Cytotec 200mcg price
September 3, 2023 at 8:08 am
prescription medication neurontin neurontin 1800 mg neurontin 100mg capsule price
September 3, 2023 at 9:22 am
Cytotec 200mcg price buy cytotec pills online cheap buy cytotec
September 3, 2023 at 10:35 am
how much is generic neurontin neurontin 600 mg cost how much is neurontin
September 3, 2023 at 11:48 am
buy cytotec pills online cheap order cytotec online Misoprostol 200 mg buy online
September 3, 2023 at 12:25 pm
F*ckin? awesome things here. I?m very glad to see your post. Thanks a lot and i’m looking forward to touch you. Will you please drop me a mail?
September 3, 2023 at 1:01 pm
ivermectin price uk stromectol drug buy stromectol
September 3, 2023 at 2:14 pm
stromectol without prescription ivermectin 1mg where to buy ivermectin
September 3, 2023 at 3:29 pm
cytotec abortion pill п»їcytotec pills online buy misoprostol over the counter
September 3, 2023 at 4:45 pm
neurontin 50 mg neurontin india neurontin 150 mg
September 3, 2023 at 9:44 pm
cost of ivermectin 1% cream ivermectin usa ivermectin price comparison
September 3, 2023 at 10:57 pm
stromectol prices stromectol in canada ivermectin cream 1%
September 4, 2023 at 12:05 am
ivermectin cost ivermectin generic ivermectin cost
September 4, 2023 at 2:23 am
order cytotec online cytotec buy online usa cytotec pills buy online
September 4, 2023 at 3:38 am
п»їcytotec pills online п»їcytotec pills online order cytotec online
September 4, 2023 at 4:55 am
stromectol pill ivermectin cream 1% ivermectin uk coronavirus
September 4, 2023 at 6:10 am
Cytotec 200mcg price buy cytotec purchase cytotec
September 4, 2023 at 7:23 am
ivermectin lotion where to buy stromectol oral ivermectin cost
September 4, 2023 at 8:35 am
ivermectin cream stromectol where to buy stromectol 6 mg dosage
September 4, 2023 at 9:49 am
ivermectin generic name stromectol 3mg cost ivermectin tablets uk
September 4, 2023 at 10:23 am
Heya i?m for the first time here. I came across this board and I find It truly useful & it helped me out much. I am hoping to offer one thing back and help others like you helped me.
September 4, 2023 at 11:00 am
gabapentin 600 mg neurontin cost neurontin brand name 800 mg
September 4, 2023 at 12:19 pm
order cytotec online buy cytotec Abortion pills online
September 4, 2023 at 1:35 pm
ivermectin 3mg tablets how much does ivermectin cost ivermectin 90 mg
September 4, 2023 at 2:51 pm
stromectol drug cost of ivermectin lotion stromectol 3 mg tablet
September 4, 2023 at 4:05 pm
neurontin 50mg cost discount neurontin neurontin 600mg
September 4, 2023 at 9:03 pm
cytotec pills buy online buy cytotec over the counter buy cytotec online fast delivery
September 4, 2023 at 11:34 pm
stromectol for humans stromectol order online ivermectin 50 mg
September 5, 2023 at 12:47 am
ivermectin where to buy buy ivermectin cream stromectol
September 5, 2023 at 2:02 am
ivermectin tablets uk ivermectin 5 stromectol price us
September 5, 2023 at 3:16 am
ivermectin 5 mg price ivermectin 500ml ivermectin cost uk
September 5, 2023 at 4:30 am
neurontin prescription cost neurontin 100 mg cost neurontin mexico
September 5, 2023 at 5:46 am
Misoprostol 200 mg buy online purchase cytotec buy cytotec online fast delivery
September 5, 2023 at 7:01 am
ivermectin rx ivermectin lotion price stromectol 0.5 mg
September 5, 2023 at 8:07 am
Spot on with this write-up, I really assume this website wants way more consideration. I?ll in all probability be again to learn much more, thanks for that info.
September 5, 2023 at 8:14 am
buy cytotec pills online cheap cytotec buy online usa buy cytotec online
September 5, 2023 at 9:31 am
buy cytotec online buy cytotec online cytotec pills buy online
September 6, 2023 at 7:05 am
Hey just wanted to give you a quick heads up. The words in your article seem to be running off the screen in Firefox. I’m not sure if this is a format issue or something to do with internet browser compatibility but I figured I’d post to let you know. The design and style look great though! Hope you get the issue resolved soon. Many thanks
September 6, 2023 at 9:59 pm
woh I am thankful to find this website through google.
September 7, 2023 at 12:44 am
I was suggested this blog by my cousin. I’m not sure whether this post is written by him as nobody else know such detailed about my trouble. You’re incredible! Thanks!
September 7, 2023 at 5:19 am
As I site possessor I believe the content material here is rattling fantastic , appreciate it for your efforts. You should keep it up forever! Good Luck.
October 3, 2023 at 5:27 am
nice and thanks